Significant Reduction in Migraine Days vs. Placebo
Meets primary and secondary efficacy endpoints
New treatment option for needle phobia or injection avoidance patients
Handok Teva’s Ajovy (containing Fremanezumab) and Eli Lilly’s Emgality (containing Galcanezumab) dominate the migraine preventive treatment market in South Korea. AbbVie Korea's introduction of Aquipta as a new oral alternative, rather than injectables, is poised to offer a novel treatment choice for both medical professionals and patients. Ajovy and Emgality currently stand as the reimbursed migraine preventive treatments available in the South Korean market, with last year's sales reaching $3.5 million and $4.5 million, respectively, according to pharmaceutical statistics data from IQVIA.
Emgality, launched in December 2019, gained prominence as the first medication specifically developed for migraine prevention. Notably, in the global clinical trial EVOLVE-2, including Korean participants, 59% of the Emgality group experienced a reduction of over 50% in the number of migraine days per month over six months.
Ajovy received approval from the Ministry of Food and Drug Safety (MFDS) in July 2021 and was listed on the reimbursement list in January 2023. Clinical trials involving over 2000 participants with episodic or chronic migraines showed significant reductions in migraine days per month in both quarterly and monthly dosing groups of Ajovy compared to the placebo group, meeting the primary efficacy endpoint.
Aquipta's entry into the migraine treatment market, previously dominated by injectable options, presents a new avenue for patients who have reservations about traditional injectables. As the only oral medication among the "calcitonin gene-related peptide (CGRP) receptor antagonists" for adult migraine prevention, Aquipta offers once-daily administration.

Dr. Byung-Geon Kim, a professor at Nowon Eulji Medical Center's Neurology Department, commented, "For patients hesitant about traditional injectables, Aquipta provides a new treatment option." He emphasized, "With minimal side effects even with daily intake, Aquipta eliminates the inconvenience of maintaining a patient medication diary, typically required for monitoring injectable treatments, thus enhancing patient convenience."
However, some suggest that improvements in reimbursement criteria should precede the widespread adoption of Aquipta, considering the stringent eligibility requirements for reimbursed treatments. It's estimated that less than 10% of patients meet the current criteria.
According to existing reimbursement criteria, eligible candidates must meet three conditions: a minimum of one year of migraine history, with at least 15 headache days per month for the past six months, including at least 8 days of migraine-type headaches per month; a scoring of 21 points or higher on the Migraine Disability Assessment (MIDAS) or 60 points or higher on the Headache Impact Test (HIT-6) before starting treatment; and failure with three or more migraine preventive medications within the past year.
Patients must also submit and maintain a headache diary for the recent six months. Failure with oral medications is defined as using each medication at the maximum daily dose for at least 8 weeks without a reduction of at least 50% in monthly migraine days or if the medication causes intolerable side effects or contraindications. Treatment duration is limited to a maximum of 12 months.
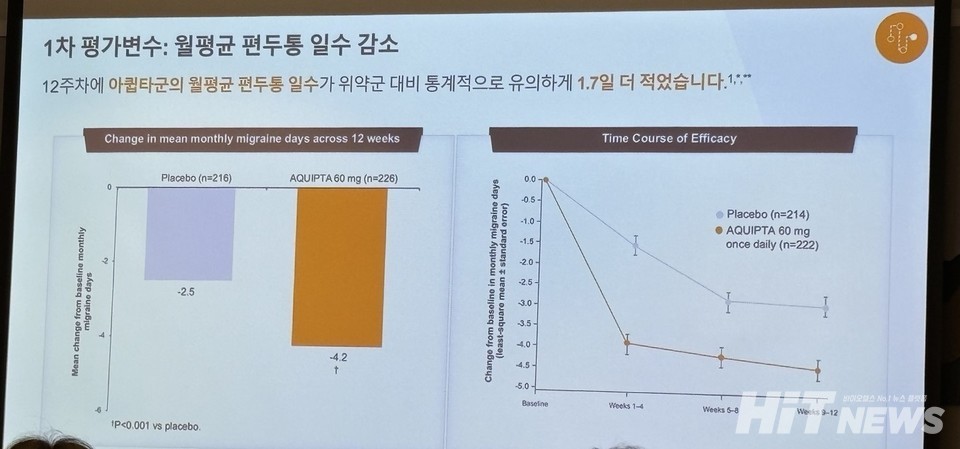
In the 'ADVANCE' clinical trial with participants suffering from episodic migraines, Aquipta demonstrated a significant reduction of 1.7 days in the average number of migraine days per month compared to the placebo group at the 12th week, meeting the primary efficacy endpoint (P<0.001) in the group receiving a daily dosage of 60 mg.
Similarly, in the 'PROGRESS' study targeting chronic migraine patients, the same dosage group exhibited superior results compared to the placebo group, with a reduction of 6.9 days in the average number of migraine days per month at the 12th week, achieving a decrease of 5.1 days (P<0.0001). Secondary efficacy endpoints were also met with a reduction of over half in headache days and acute medication usage days.

