Tovetisomig Cuts Cholangiocarcinoma Mortality
Givastomig Finishes Early Phase 1b Enrollment
ABL Bio Speeds Pipeline Diversification

ABL Bio’s bispecific antibody pipeline is gaining momentum, with two lead programs—tovetisomig and givastomig—showing encouraging progress in global clinical trials. Tovetisomig, a second-line therapy candidate for cholangiocarcinoma, has demonstrated potential survival benefits, while givastomig is moving ahead of schedule in gastric cancer development.
On August 11, ABL Bio said its partners Compass Therapeutics (Nasdaq: CMPX) and I-Mab Therapeutics (Nasdaq: IMAB) released new clinical updates for the two drug candidates. Key efficacy data are expected in the first half of 2026, which could accelerate commercialization plans.
‘Tovetisomig’ Extends Survival in Cholangiocarcinoma
Tovetisomig (ABL001, VEGF-A x DLL4), developed on ABL Bio’s Grabody-T platform and licensed to Compass Therapeutics, is being evaluated in the Phase 2/3 COMPANION-002 trial for advanced cholangiocarcinoma. Interim results suggest a survival benefit, prompting a delay in final analysis until early 2026 due to fewer-than-expected death events.
In topline data released in April, the combination of tovetisomig and paclitaxel achieved an objective response rate (ORR) of 17.1%, more than triple the 5.3% seen with paclitaxel alone (p=0.031), including one complete response. The trial is designed to assess overall survival (OS) and progression-free survival (PFS) once 80% of targeted death events occur.
Tovetisomig targets both VEGF-A and DLL4–Notch pathways to inhibit tumor angiogenesis and prevent compensatory signaling. This dual blockade aims to enhance anti-tumor activity, with an early “vascular normalization” phase improving chemotherapy delivery.
Previous Korean Phase 2 results showed an ORR of 37.5% overall—63.6% in second-line and 15% in third-line settings—median PFS of 9.4 months, and a one-year survival rate of 53%, outperforming historical FOLFOX benchmarks. Compass plans a Biologics License Application in mid-2026 and will pursue basket trials in DLL4-high tumors, including gastric, ovarian, and liver cancers.
‘Givastomig’ Moves Faster in Gastric Cancer Program
Givastomig (ABL111, Claudin18.2 x 4-1BB), co-developed with I-Mab, has completed patient recruitment for the Phase 1b dose-expansion trial in Claudin18.2-positive gastric cancer ahead of schedule. Forty patients were enrolled to test 8 mg/kg and 12 mg/kg doses, both of which showed acceptable safety.
The trial combines givastomig with Opdivo (nivolumab) and mFOLFOX6 chemotherapy as first-line treatment. This regimen uses chemotherapy to create a favorable immune environment, followed by givastomig to activate 4-1BB and nivolumab to sustain T-cell activity.
Unlike Astellas’ Zolbetuximab, which is limited to patients with ≥75% Claudin18.2 expression, givastomig is designed for tumors with 1–100% expression, potentially doubling the eligible population. Earlier trials showed an ORR of 83% in active dose groups, with tumor shrinkage in 10 of 12 patients.
Platform Expansion, Pipeline Diversification to Accelerate Growth
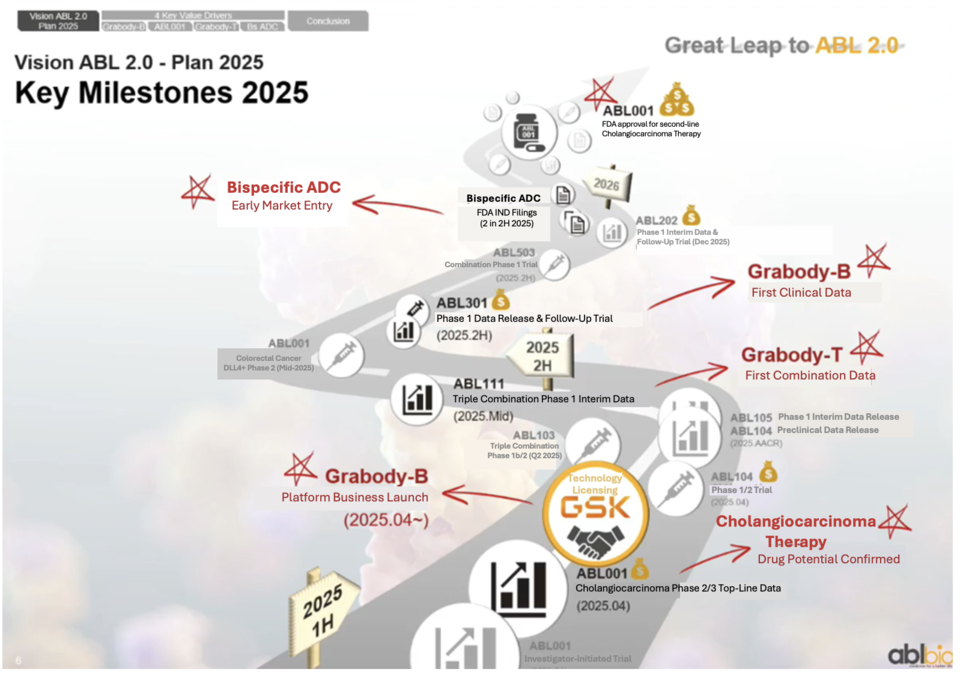
Beyond Grabody-T, ABL Bio’s portfolio includes the Grabody-B BBB shuttle platform—licensed to Sanofi and GSK—antibody-drug conjugates (ADCs), and multi-target bispecific antibodies. Grabody-B is under exploration for Alzheimer’s disease, other neurodegenerative conditions, siRNA-based muscle therapies, and obesity treatments.
The oncology pipeline includes ROR1 ADC (ABL202), α-synuclein-targeting ABL301, PD-L1 x 4-1BB (ABL503), HER2 x 4-1BB (ABL105), B7-H4 x 4-1BB (ABL103), and EGFR x 4-1BB (ABL104).
An ABL Bio spokesperson said, “Survival data for ABL001 are promising, as patients are living longer than anticipated. With accelerated development of ABL111, we also plan to advance combination trials for our other 4-1BB bispecific antibodies.”

