Onbevezy and Samfenet sales surge, positioning biosimilars at the core of Boryung’s oncology franchise
Boryung’s biosimilar strategy is gaining momentum, with sales of its oncology biosimilars rapidly surpassing older drugs to become the company’s new growth drivers.
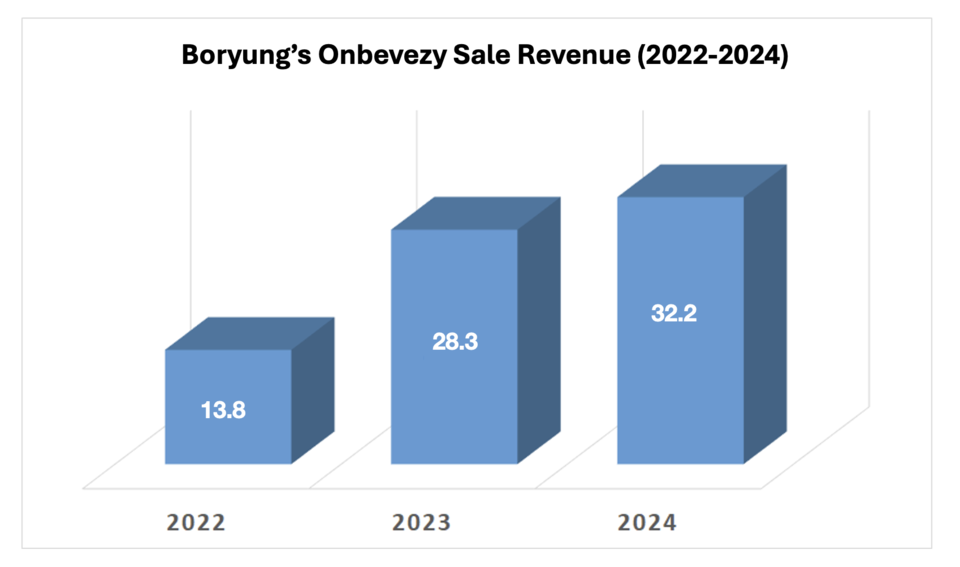
According to company disclosures on September 29, sales of the Avastin biosimilar Onbevezy jumped from $13.8 million in 2022 to $32.2 million in 2024. Boryung in-licensed the product from Samsung Bioepis in 2021, securing exclusive South Korean rights.
Industry observers attribute the success to Boryung’s scale in oncology. With Korea’s largest oncology salesforce and a portfolio of more than 30 anticancer drugs—including Taxol—the addition of Onbevezy created strong synergies.
Onbevezy is prescribed for colorectal and gynecological cancers, areas where Boryung already markets Campto, Oxalitin, and Taxol. A medical expert noted:
“Taxol has long been a first-line treatment, but once resistance develops, Onbevezy provides a new option. This complementarity likely boosted Boryung’s sales effectiveness.”
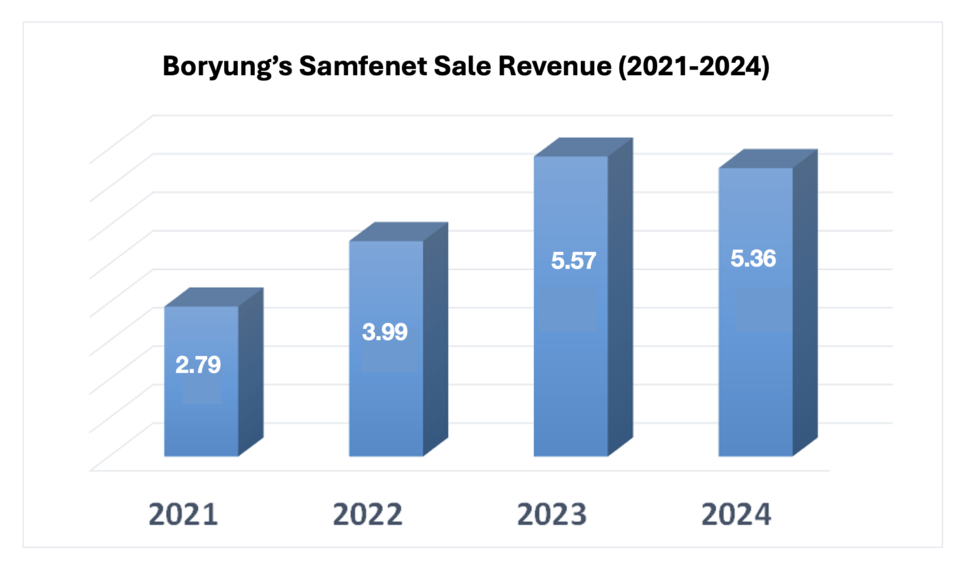
Boryung’s second biosimilar, Samfenet, a Herceptin biosimilar, has also shown steady growth. Sales nearly doubled since launch, rising from $2.8 million in 2021 to $5.4 million in 2024. Licensed from Samsung Bioepis in December 2021, Samfenet benefited from synergies with Boryung’s breast cancer drug Fulvet.
By 2024, Onbevezy overtook long-standing supportive therapy Neulasta ($21.8 million) to become Boryung’s top oncology product.
The dual success of Onbevezy and Samfenet reflects the company’s “Five-Year Mid-to-Long Term Plan,” initiated in 2022, which set a goal of growing oncology sales from $71 million to $500 million.
A South Korean oncology expert explained: “Boryung’s past strategy centered on acquiring rights to mature originals like Alimta and Gemzar. These are proven drugs, but their growth potential is limited. Biosimilars, by contrast, are entering markets already proven in scale, with adoption accelerating under cost-containment policies in the U.S., Europe, and Korea.”
In July, Boryung expanded its pipeline by in-licensing Xbryk, a biosimilar of Xgeva for bone disorders—its third oncology biosimilar with Samsung Bioepis.
An industry source observed: “Biosimilars match the efficacy of originals, so execution is everything. Development is complex, but without strong commercialization, they fail. Boryung has unmatched oncology sales expertise, and if Xbryk follows the trajectory of Onbevezy and Samfenet, biosimilars will become the centerpiece of its next-generation growth strategy.”

