Government and Industry to Collaborate on Revising Price Adjustment Mechanism for Expanding Drug Indications

The "Indication Expansion Negotiation System," which mandates price reductions when drug indications are expanded, is set for revision within the year. The government and the pharmaceutical industry plan to establish a consultative body to discuss improvement measures.
According to industry sources on February 19th, the National Health Insurance Service (NHIS) and the pharmaceutical and biotech sectors will form a joint working group to review potential system revisions. Representatives from the Korea Pharmaceutical and Bio-Pharma Manufacturers Association, the Korea Research-based Pharma Industry Association, and the Korea Biopharmaceuticals Association will participate in the discussions.
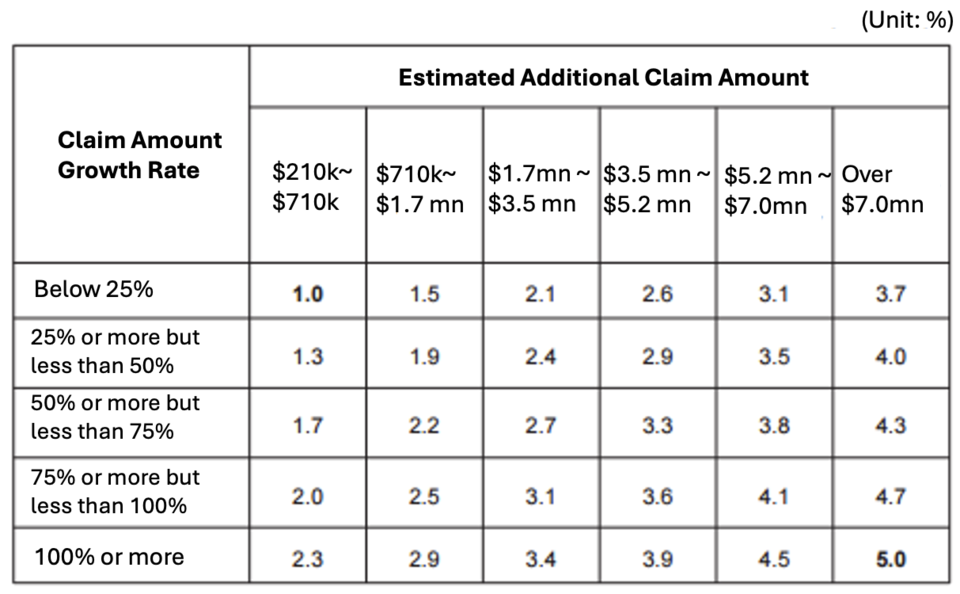
The NHIS previously conducted a study on improving the system, driven by concerns that high-cost drugs, including immuno-oncology treatments, often receive early approval and reimbursement, increasing financial strain on the national health insurance system. Despite this growing burden, substantial reforms had yet to be implemented.
Introduced in 2014, the system has remained unchanged for over a decade. A study by Professor Eun-A Han of Yonsei University’s College of Pharmacy suggested short-term reforms such as revising criteria for selecting negotiation targets and adjusting the price reduction formula. The study also recommended clearer guidelines for negotiation procedures.
For mid-to-long-term improvements, the study proposed incorporating clinical evidence and cost-effectiveness assessments into financial impact evaluations. Other recommendations included long-term financial impact assessments to inform pricing adjustments, a structured management framework for multi-indication drugs, and a potential total budget system.
Pharmaceutical industry stakeholders argue that the current system lacks predictability, as it mandates price reductions in anticipation of increased claims and growth rates. They emphasize the need for safeguards to prevent excessive alignment with volume-based pricing frameworks.
Industry representatives have proposed adjusting the criteria for preemptive price reductions to apply only to drugs with expenditure increases between $34.8 million and $348 million. They also advocate for a comprehensive post-management system.
An industry official stated, "While no specific meeting dates have been set, the plan is to complete the system reform within the year based on research findings. Instead of fragmented adjustments, such as changes to the actual transaction price reduction system, a more integrated post-management approach is necessary."

