Seeks Approval for 17 Indications, Including Gastric Cancer, as Severe Disease Committee Review Looms

Korea MSD has submitted a financial-sharing proposal to expand reimbursement criteria for Keytruda (pembrolizumab) and awaits a decision by the Severe Disease Deliberation Committee in December. This follows the first expansion of reimbursement criteria for non-small cell lung cancer in five years, raising questions about how long the current process will take.
According to industry sources on November 22, Korea MSD submitted supplementary materials in October to the Health Insurance Review and Assessment Service (HIRA), covering financial-sharing proposals for 17 indications, including gastric cancer. Earlier, the company applied in June 2023 for expanded reimbursement of 13 indications, such as triple-negative breast cancer, head and neck cancer, and esophageal cancer. Additional applications for four indications, including MSI-H bile duct cancer and gastric cancer, followed at the end of 2023 and February 2024.
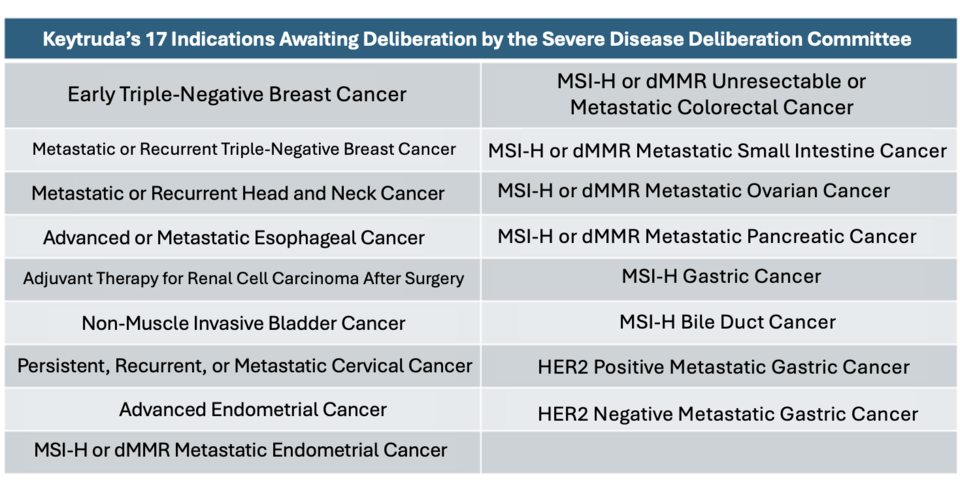
In total, reimbursement applications for 17 indications have been submitted, but a reevaluation ruling in April delayed deliberations by the Severe Disease Committee. Among the pending indications, gastric cancer, submitted immediately after approval, has yet to undergo its initial review. If the committee meeting scheduled for December 18 fails to approve the application, Keytruda's reimbursement request will enter its third year.
Korea MSD faces further challenges due to limitations that prevent new indications approved after the initial submission from being included in the reimbursement request. The company has highlighted the urgent need for committee review, particularly for indications like gastric cancer, triple-negative breast cancer, and head and neck cancer, where no reimbursed cutting-edge therapies currently exist.
Korea MSD submitted updated financial-sharing proposals in October and emphasized the importance of timely reimbursement during the National Assembly’s Health and Welfare Committee audit this year. Representative Jumin Park (Democratic Party) underscored the significance of expanding access to immuno-oncology treatments like Keytruda for Korean patients.
A Korea MSD representative stated, “We aim to provide treatment benefits to patients by addressing the unique challenges of immuno-oncology drugs with multiple indications. We are ready to engage in institutional discussions following the committee’s decision. Our goal is to secure a review of Keytruda this year, improving access to life-saving treatments for cancer patients in Korea.”

