AstraZeneca launches AKT inhibitor 'Tiruqap' at press conference
Prof. Kyung-Hwa Park: "Only second-line option for patients on CDK inhibitors"
Despite advancements in hormone receptor-positive (HR+) breast cancer treatments, including a 5-year survival rate with oral medications, resistance remains a challenge due to cellular changes, gene expression, and mutations.
To address this, the AKT inhibitor 'Tiruqap' (capivasertib) has been introduced for patients who develop resistance after first-line treatment with cyclin-dependent kinase (CDK) inhibitors and endocrine therapy. Korea AstraZeneca held a press conference on September 12th at Oakwood Premier Coex Center to launch Tiruqap.

Professor Kyung-Hwa Park of the Oncology Department at Korea University Anam Hospital explained that mutations such as ESR1 and PIK3CA/AKT often arise after first-line treatments with CDK4/6 inhibitors in HR-positive/HER2-negative breast cancer, which represents 70% of breast cancer cases. Notably, PIK3/AKT pathway mutations are found in 40-50% of HR-positive patients.
Professor Park noted that while HR-positive breast cancer typically metastasizes to bones, resistance to endocrine therapy often leads to metastasis in internal organs such as the liver. Two-thirds of patients experience internal organ metastasis, with 40% facing poor prognoses.
"For patients treated with CDK inhibitors, avoiding cytotoxic chemotherapy is crucial to maintaining their daily life and quality of life," said Professor Park. "For these patients, Tiruqap combined with fulvestrant is the only available AKT inhibitor therapy for second-line treatment."
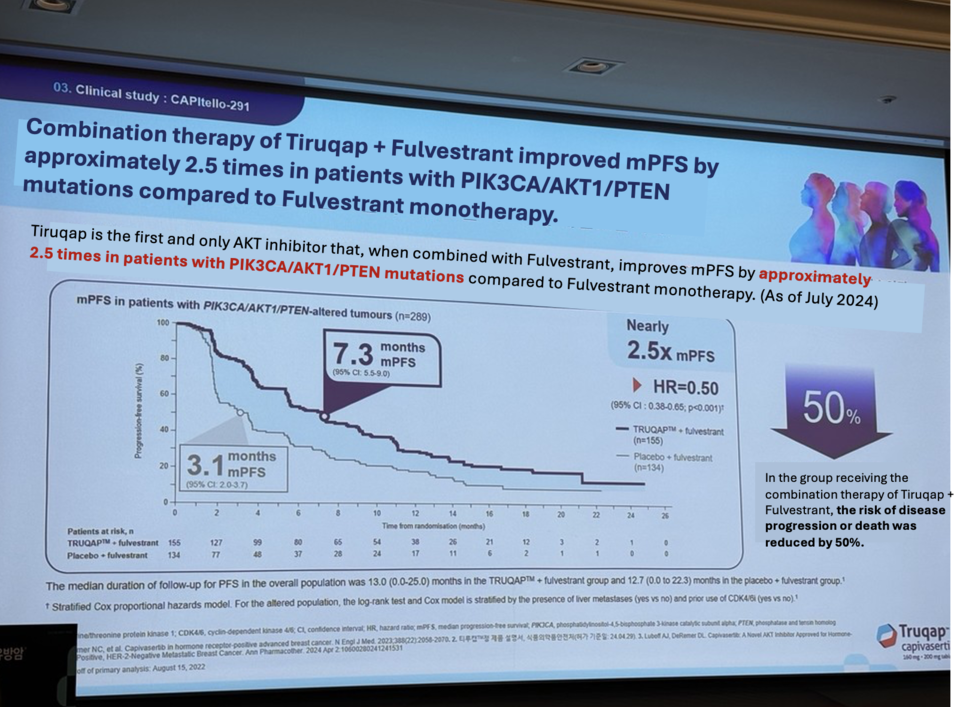
Phase 3 clinical trial results from the 'CAPItello-291' study showed that Tiruqap improved median progression-free survival (mPFS) to 7.3 months, 2.5 times longer than the control group's 3.1 months. It also reduced the risk of disease progression and death by 50% and improved overall survival (OS) by 30% compared to monotherapy.
Professor Park noted, "The Ministry of Food and Drug Safety (MFDS) has approved Tiruqap for patients with gene mutations like PIK3CA, AKT1, or PTEN. However, detecting these mutations requires Next-Generation Sequencing (NGS), which is often inaccessible due to financial costs, as patients must cover over 80% of the cost."
She added, "NGS is essential for patients to have the option to choose their treatment. While Tiruqap is now available, the next hurdle is securing insurance coverage for NGS testing."

Professor Park emphasized, "While Tiruqap offers hope for 50% of patients with gene mutations, this hope is contingent upon NGS testing, which can reduce the financial burden on patients."
Jae-Yoon Im, Executive Director of Medical Affairs at AstraZeneca Korea, also outlined the company’s breast cancer strategy. Im highlighted the importance of utilizing diverse anticancer mechanisms, promoting early detection, and leading innovative technologies in breast cancer treatment.
He added, "Currently, the 5-year survival rate is 86% for early-stage breast cancer and 30% for metastatic cases. Our R&D aims to improve these rates to 90% and 50%, respectively, by 2030."

