Expanded Reimbursement for First-Line Lung Cancer Treatment
8-Month Prescription Revenue Matches Last Year’s Total
The EGFR mutation-targeted therapies for non-small cell lung cancer (NSCLC), Tagrisso (ingredient: Osimertinib) and Leclaza (ingredient: Lazertinib), have shown concurrent growth following the expansion of first-line reimbursement criteria.
According to pharmaceutical research firm UBIST, AstraZeneca's Tagrisso recorded $61.7 million in prescription revenue up to August this year. Prescription revenue steadily increased from $53.4 million in 2020 to $58.6 million in 2021 and $67.5 million in 2022. However, the introduction of South Korea's Leclaza into clinical practice led to a slight dip in Tagrisso's revenue, down to $66.8 million in 2023.
Following the expansion of its indication for first-line lung cancer treatment in January, Tagrisso saw a significant surge in revenue. Cumulative revenue from January to August has already reached $61.7 million, nearly matching the total from last year. The drug’s price reduction has also encouraged its use as adjuvant therapy in early-stage EGFR-mutated NSCLC patients after surgery. With four months remaining in the year, Tagrisso's total revenue is expected to surpass $74.7 million.
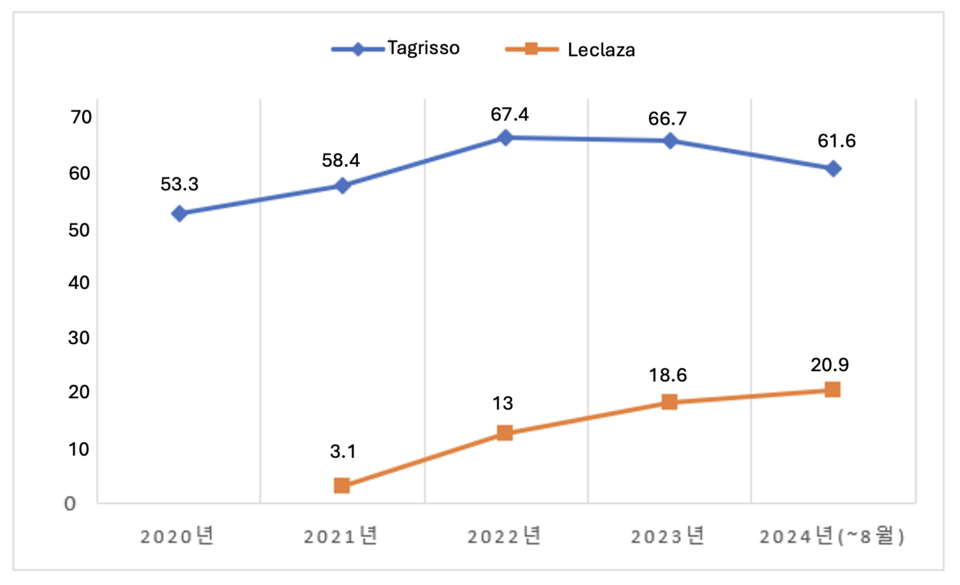
Leclaza has also experienced rapid growth, with prescription revenue climbing from $3.1 million in 2021 to $13 million in 2022 and $18.7 million in 2023. From January to August this year, Leclaza has already generated $21 million, exceeding last year's total.
This growth is largely attributed to the expanded reimbursement for first-line lung cancer treatment, initiated in January. Furthermore, Leclaza has gained attention for its combination therapy with Rybrevant (ingredient: Amivantamab). A study presented at the World Conference on Lung Cancer (WCLC 2024) showed that patients with EGFR-mutated NSCLC (exon 19 deletion or L858R) achieved higher 3-year progression-free survival rates with the Rybrevant + Leclaza combination compared to Tagrisso monotherapy.
At 24 months, survival rates were 75% (95% CI: 71-79) for the Rybrevant + Leclaza group, compared to 70% (95% CI: 65-74) for the Tagrisso group. By 36 months, the gap widened further, with survival rates of 61% (95% CI: 56-67) for the combination therapy and 53% (95% CI: 47-59) for Tagrisso.
On August 20, the FDA approved the Rybrevant + Leclaza combination therapy. Further data from the MARIPOSA-2 trial, comparing the combination of Rybrevant and chemotherapy against chemotherapy alone in EGFR-mutant advanced NSCLC patients who have progressed after Tagrisso treatment, will be presented at the European Society for Medical Oncology (ESMO 2024) conference on September 13.

