FLAURA2 Asian Subgroup Analysis to Guide Optimal Treatment for Asian Patients
Tagrisso Excels in Brain Metastasis Prevention and Quality of Life
The treatment landscape for non-small cell lung cancer (NSCLC) is advancing rapidly, particularly with third-generation tyrosine kinase inhibitors (TKIs) like Tagrisso (osimertinib). Since its launch, Tagrisso has been used by over 700,000 patients worldwide, transforming the management of EGFR-mutated NSCLC into a more chronic disease-like condition. In South Korea, it was approved in 2016 as a second-line therapy for EGFR-T790M mutation-positive NSCLC and expanded in 2018 as a first-line therapy for locally advanced or metastatic NSCLC with EGFR mutations.
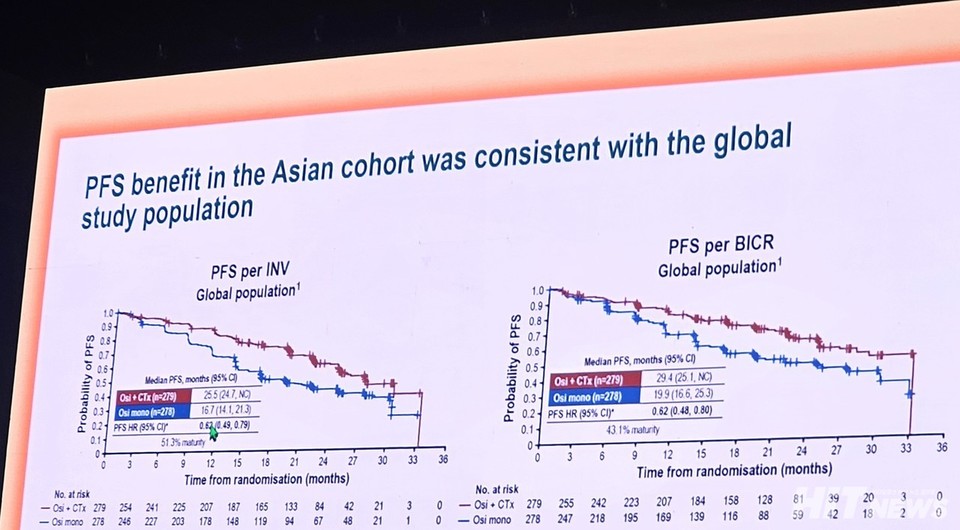
The global Phase III FLAURA2 trial highlights Tagrisso’s role as a first-line therapy when combined with chemotherapy, especially for patients with EGFR exon 19 deletions or exon 21 (L858R) substitution mutations. At the ESMO Asia 2024 Annual Congress, the Asian subgroup analysis from this trial drew significant attention, reflecting the unique prevalence of lung cancer in Asian populations and the importance of tailored treatment strategies.
Hit News explored the clinical value of Tagrisso combination therapy, expanded through the FLAURA-2 study, and its impact on the global NSCLC treatment paradigm since the drug’s introduction. Dr. Daniel Chan of Singapore’s Icon Cancer Centre shared his insights on these developments during an on-site interview at ESMO Asia 2024.
What is the significance of the Asian subgroup analysis from the FLAURA2 study?
"EGFR-mutated NSCLC is far more common in Asians than in Western populations, making this subgroup analysis particularly important. Asian researchers have substantial experience with these patients, enabling them to conduct high-quality studies. The FLAURA2 analysis highlights differences in treatment outcomes for Asian patients compared to global data, offering insights into optimizing therapy. For instance, earlier studies, like FLAURA's Japanese subgroup analysis, revealed how variables like adverse event profiles can differ between populations. This new data will help refine treatment guidelines for Asian patients."
How has the treatment environment for patients improved before and after the launch of Tagrisso?

"Tagrisso has been transformative in the treatment of lung cancer, evolving what was once considered an incurable disease into something more manageable, like hypertension or diabetes. When Tagrisso was first introduced in Singapore in 2015 through a patient support program, its efficacy was immediately evident, even in patients with brain metastases—a challenge that earlier EGFR-TKIs struggled to address effectively.
Unlike first- and second-generation EGFR-TKIs, which offered limited protection against brain metastases, Tagrisso has proven highly effective in both protecting the brain and preventing metastases. Moreover, patients treated with Tagrisso have shown remarkable improvements in maintaining their quality of life. Many are able to continue working and fulfilling family responsibilities during treatment, which marks a significant shift from the limitations imposed by previous therapies.
This impact is also recognized in the National Comprehensive Cancer Network (NCCN) guidelines, where Tagrisso monotherapy is the only recommended first-line treatment option. Such recognition underscores its value not only in extending survival but also in preserving patients' quality of life."
What do you think are the criteria for determining suitable patient groups for Tagrisso combined with chemotherapy?
"The FLAURA2 study demonstrated that the combination of Tagrisso with chemotherapy offers significant survival benefits, particularly for high-risk patients with a substantial disease burden. At the recent World Conference on Lung Cancer (WCLC), an updated analysis from FLAURA2 identified patients with a high disease burden based on the presence of three or more metastatic sites.
For these patients, the hazard ratio for median progression-free survival (PFS) was reported as 0.57, compared to 0.75 in those with fewer than three metastatic sites. This analysis, though not initially planned, clearly shows that combination therapy is more effective in patients facing a heavier disease burden.
While additional insights may emerge from the Asian subgroup analysis of FLAURA2, it is already evident that combination therapy is particularly beneficial for patients with poor overall health or advanced disease. These findings will play a critical role in refining treatment strategies for this population."
Despite the higher rate of grade 3 or higher adverse events in the Tagrisso combination group, the proportion of patients discontinuing Tagrisso treatment was not significantly different. How should this be interpreted?
"The adverse events reported in the FLAURA2 study are primarily linked to the cytotoxic chemotherapy agents used in combination therapy, not to Tagrisso itself. For example, anemia—a major adverse event—is caused by the platinum-based agent carboplatin, while neutropenia and thrombocytopenia are also associated with chemotherapy.
To address these issues, the study allowed for dose adjustments or reductions in chemotherapy to ensure patient safety. If more than two dose modifications were needed, the protocol allowed for discontinuing the chemotherapy. This flexibility likely enabled patients to continue Tagrisso treatment safely, even when experiencing grade 3 or higher adverse events.
Importantly, adding chemotherapy did not worsen Tagrisso’s known side effects or introduce any new adverse effects specific to Tagrisso, ensuring its safety profile remained consistent."
At this year’s WCLC, a subgroup analysis from the MARIPOSA study showed a longer median progression-free survival (PFS) for the Leclaza + Rybrevant combination therapy compared to Tagrisso monotherapy. How should we interpret this data?
"It is essential to analyze the entire survival curve rather than focusing solely on isolated figures like median PFS. Median PFS reflects the time at which disease progression is observed in the 51st patient in a group of 100, but it does not capture the overall trend of patient responses across the cohort. The broader curve provides a more comprehensive understanding of treatment effectiveness.
When comparing studies, the clinical effects of Tagrisso combined with chemotherapy and the Leclaza + Rybrevant combination appear to be quite similar. However, directly comparing the median PFS values of the lazertinib and Tagrisso monotherapy groups from the MARIPOSA study’s subgroup analysis is not appropriate without considering the entire survival graph.
The confidence intervals for the median PFS of both drugs overlap significantly. For Tagrisso, the interval is 14.8 to 18.5 months, while for Leclaza, it is 14.8 to 20.1 months. This overlap suggests that random variability may account for the observed differences.
Leclaza is undoubtedly an excellent drug in clinical practice. However, claiming superiority over Tagrisso based solely on this data would not be scientifically sound. A fair assessment requires a more nuanced interpretation of the study results."
What direction do you think the development of EGFR-mutated lung cancer treatments will take in the future?

"The development of second-line therapies for patients who experience disease progression after Tagrisso will likely be a key focus over the next 5–10 years. Currently, many patients in this situation rely on treatments like radiation or other localized therapies, but the available options beyond these are limited and have not shown remarkable efficacy.
For instance, standard chemotherapy provides a survival benefit of about five months. Similarly, the MARIPOSA-2 study, which combined Rybrevant with chemotherapy, reported a progression-free survival (PFS) of 6.3 months. While these results represent incremental progress, they are far from groundbreaking. AstraZeneca’s antibody-drug conjugate (ADC), datopotamab deruxtecan (Dato-DXd), has shown promise but only in certain patient subgroups.
In this context, AstraZeneca’s upcoming TROPION-Lung15 trial is particularly noteworthy. This Phase III study, set to begin in January, will evaluate the efficacy of three approaches—Tagrisso combined with Dato-DXd, Dato-DXd monotherapy, and standard chemotherapy—in patients who have failed Tagrisso monotherapy. The trial is expected to provide critical insights into the most effective treatment strategies for this patient group.
That said, the treatment landscape may evolve significantly before the trial concludes. If ongoing studies like FLAURA2 and MARIPOSA demonstrate overall survival (OS) benefits for high-risk patients, combination therapies could become the new standard for first-line treatment. This shift might reduce the focus on second-line options for patients initially treated with monotherapy."

