Shin & Kim Holds Briefing on Lawsuit to Nullify Reimbursement Contracts for Pharma Companies
Uncertainty in Clinical Re-evaluation Results Raises Concerns
Some Companies May Join the Lawsuit if Costs Are Low

A new spotlight is being cast on whether a lawsuit will be filed to nullify the reimbursement contract between the National Health Insurance Service (NHIS) and pharmaceutical companies related to choline alfoscerate preparations.
According to industry insiders, the law firm Shin & Kim hosted a briefing in July for pharmaceutical companies involved in the choline reimbursement negotiations. The briefing explored the possibility of invalidating the existing contract, with the argument focusing on preventing financial losses that could arise from the current reimbursement terms.
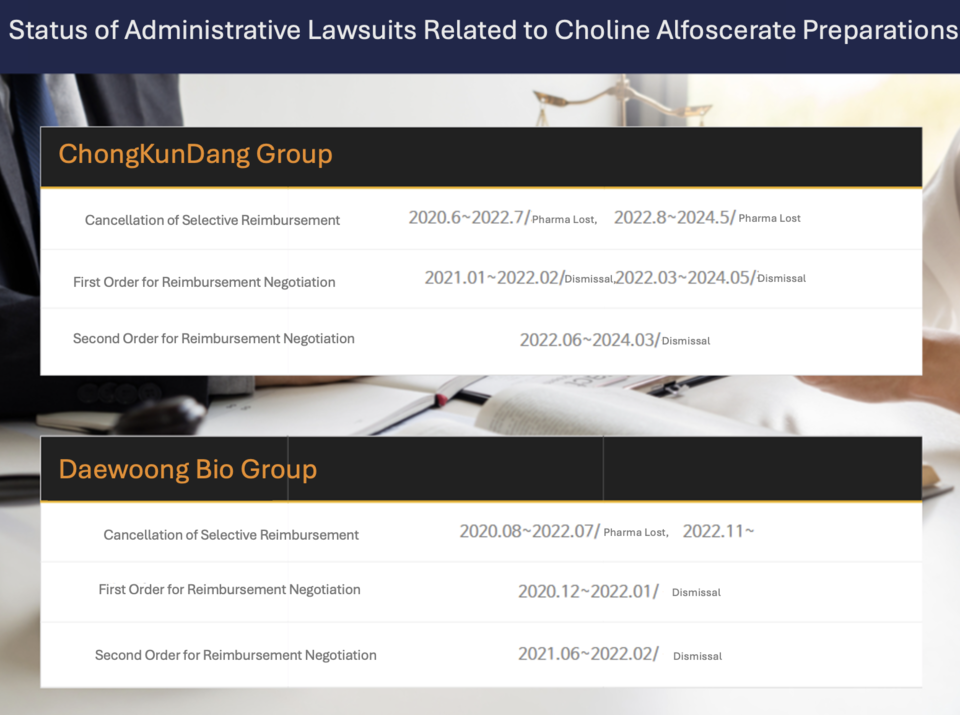
Legal battles concerning choline preparations have so far revolved around two primary areas: lawsuits seeking to overturn the selective reimbursement notice and those contesting the reimbursement negotiation orders. The government has prevailed in all related cases thus far. In August 2020, the Ministry of Health and Welfare (MOHW) implemented selective reimbursement for indications outside of dementia, prompting pharmaceutical companies to challenge the decision in court. Represented by the law firms Bae, Kim & Lee and Shin & Kim, these companies have faced continuous setbacks. The case is currently in its third appeal, although the selective reimbursement notice remains suspended.
The administrative lawsuits over the reimbursement negotiation orders can be divided into two phases. The first phase began when the MOHW issued an order in December 2020 directing the NHIS to recover treatment costs from pharmaceutical companies if related clinical trials failed. A similar lawsuit followed the issuance of a subsequent negotiation order in June 2021.
These lawsuits have split representation between Bae, Kim & Lee and Shin & Kim. In one prominent case, Daewoong Bio Group, represented by Bae, Kim & Lee, saw 25 companies withdraw, except for CMG Pharmaceutical and Whanin Pharmaceutical, leading to a dismissal ruling in 2022, which was not appealed. Similarly, in the case led by Shin & Kim on behalf of the Chong Kun Dang Group, a dismissal ruling was issued, and the appeal was denied, leaving the case currently under review in a third appeal.
Additionally, concern continues to grow over the clinical re-evaluation of choline preparations. The results for the Alzheimer’s indication are expected by December 2025, while those for cognitive impairment are due by March 2025. However, delays in medical services and difficulties with patient recruitment have sparked discussions about potentially extending these deadlines.
Pharmaceutical companies involved in the clinical re-evaluation are facing additional costs. Should they decide to halt the trials, they would be forced to withdraw their products, triggering reimbursement claims by the NHIS. Given this context, Shin & Kim considers this an optimal time to file a lawsuit to invalidate the reimbursement contract. The firm also pointed out that any ruling would apply only to the companies participating in the lawsuit.
A representative from one of the companies holding choline preparations commented, "We cannot be entirely confident in the outcome of the clinical re-evaluation. Since this lawsuit questions the validity of the entire negotiation process, it’s a viable option from the company’s perspective. We’ve decided to participate, especially considering the cost is not prohibitive."
Another industry source noted, "Shin & Kim seems focused on securing a definitive ruling before the clinical re-evaluation results are finalized. While they encourage as many companies as possible to join the lawsuit, each company is carefully weighing its options. For now, we are monitoring developments closely."

