‘Anbal-cel’ Phase 2: 71% Complete Remission by Year-End
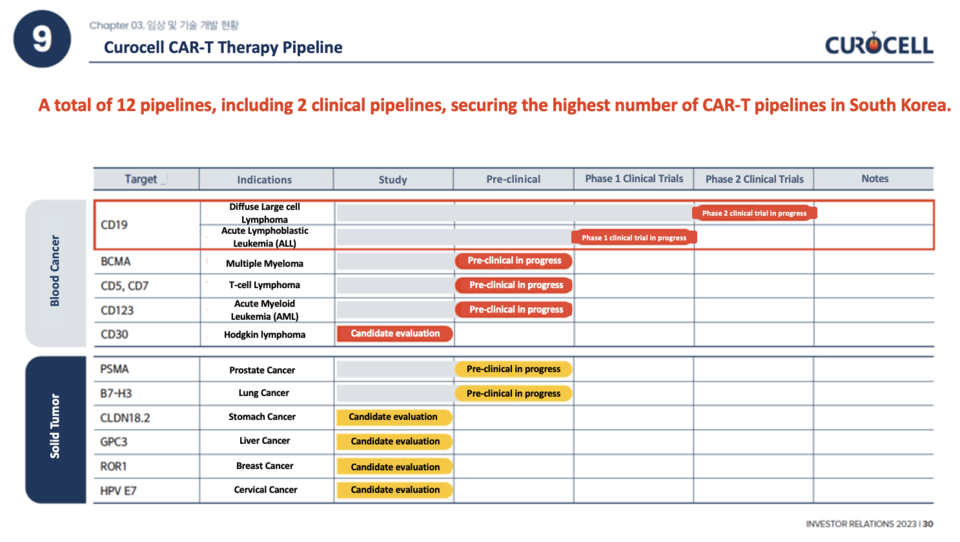
12 Pipelines, Expanding CRC01 to Adult ALL

Curocell, a biotech company founded in 2016, is actively developing Chimeric Antigen Receptor T-cell (CAR-T) therapies rather than relying on technology transfers. Their goal is to gain approval for 'Anbal-cel,' a CAR-T therapy with Anbalcabtagene Autoleucel, by submitting a New Drug Application (NDA) to Ministry of Food and Drug Safety (MFDS) in September next year, aiming for approval by 2025.
Geon-Soo Kim, Curocell's CEO, shared these plans during a press conference for their initial public offering (IPO) on October 20th in Seoul. Curocell has attracted $78.8 million in investments and is dedicated to advancing next-generation CAR-T therapies. CAR-T therapy modifies a patient's T-cells to target and eliminate cancer cells, a type of Cell and Gene Therapy (CGT).
In February 2021, Curocell received approval for its Investigational New Drug (IND) plan from MFDS, making it the first in South Korea to develop CAR-T therapy targeting CD19, called "Anbal-cel." This therapy uses 'OVIS (Overcome Immune Suppression) technology' to inhibit immune checkpoint receptors 'PD-1' and 'TIGIT,' and enhances the effectiveness of existing CAR-T therapies in the global market.
Kim stated, "Curocell aims to be a global leader in anti-cancer immune cell therapies. We're competing globally to develop innovative drugs and actively supplying anti-cancer immune cell therapies." He added, "CAR-T therapies are highly effective in advanced blood cancers, and the global competition to extend their use to solid tumors is fierce."
He continued, "Curocell will introduce South Korea's first CAR-T therapy, establishing itself as a leader in anti-cancer immune cell therapies globally. We're leveraging our advanced CAR-T technology and top-tier research and development capabilities to secure our position in the global market."
OVIS Technology enhances CAR-T and immune checkpoint effects.
Third-generation immunotherapies have emerged to overcome limitations in conventional cancer treatments and improve therapeutic outcomes. These therapies focus on enhancing the body's own immune cells to target cancer cells, in contrast to traditional drug-based approaches. Notable examples include 'CAR-T therapy' and 'immune checkpoint inhibitors.' The immunotherapy market is projected to reach approximately $126.9 billion by 2026, with CAR-T therapy involving the genetic modification of a patient's T-cells to target specific cancer cells before reintroduction into the body.
Curocell, pioneering clinical trials for CAR-T therapy in South Korea, boasts exceptional technical expertise. Their proprietary OVIS technology is highly regarded as a groundbreaking fusion technology that combines the advantages of CAR-T therapy and immune checkpoint inhibitors.
In the typical process of T-cells eliminating cancer cells, the production of immune checkpoint receptors on T-cell surfaces increases. These receptors bind to specific regions (ligands) on cancer cells, significantly impeding the T-cells' cancer-killing ability. Immune checkpoint inhibitors are designed to block the function of these receptors, thereby enhancing T-cell effectiveness in eliminating cancer cells.
CAR-T therapy also uses T-cells, and during the process of eliminating cancer cells, immune checkpoint receptor expression on CAR-T cells increases. Mr. Kim explained, "Through OVIS technology, Curocell has successfully achieved simultaneous inhibition of two immune checkpoint receptors, 'PD-1' and 'TIGIT,' while enhancing CAR expression in T-cells through genetic modification." He added, "We have filed patents for OVIS in 25 countries, including South Korea, and successfully registered patents in South Korea, the United States, Europe, Japan, and other regions."
According to the company, clinical trial samples analysis has shown that Anbal-cel reduces PD-1 expression by 70% and TIGIT by 90%, factors that can impede the effectiveness of CAR-T therapy. The company explains that these results represent a significant enhancement compared to conventional CAR-T therapies available in the global market, such as Kymriah, Breyanzi, and Yescarta.
In the interim results of its Phase 2 clinical trial, Anbal-cel achieved an outstanding complete response rate (CR) of 71%, surpassing the 40% CR rate of Novartis' 'Kymriah,' the sole CAR-T therapy available in South Korea. The company presented these results at the International Conference on Malignant Lymphoma (ICML 2023), a prestigious global lymphoma conference held in June.
Buoyed by the impressive technological prowess demonstrated by Anbal-cel, Curocell is not only expanding its indications but also actively pursuing new projects. In the realm of hematologic malignancies, they are researching pipeline candidates for six diseases, including T-cell lymphoma and CD19. In the field of solid tumors, research is underway for six diseases, including prostate cancer and lung cancer.
Anbal-cel Phase 2 completion in 2023 and South Korean NDA submission in September 2024.
Curocell's ambitious agenda includes advancing the full-scale commercialization of Anbal-cel in the coming year. This milestone follows the successful conclusion of Phase 2 clinical trials earlier in the year and the submission of a NDA in the latter part of the same year. Notably, the company has made remarkable progress in optimizing the quality inspection process for CAR-T therapies. They have introduced a 'rapid inspection method' which has drastically reduced the inspection time from 30 days to a mere 7 days. This pioneering method has been officially endorsed by the MFDS, making it the first of its kind in South Korea.
The implementation of the rapid inspection method has significantly shortened the supply timeline for Anbal-cel, making it feasible for patients to receive treatment within just 16 days from the initial collection of their immune cells. This achievement is deemed strategically advantageous, especially given the rapid progression of advanced cancer in patients.
CEO Geon-Soo Kim shared insights, stating, "Our focus has been on the development of CD19 CAR-T therapy for patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL). Curocell successfully completed Phase 1 clinical trials in 2021." He went on to elaborate, "We have recently concluded the treatment of the last patient in Phase 2 clinical trials, and based on these promising results, we intend to submit an NDA to the MFDS in September of the coming year."
Additionally, the company has expanded its scope to encompass an additional indication for CRC01 (Anbal-cel), specifically in the treatment of adult acute lymphoblastic leukemia (ALL). Their strategic plan involves submitting an NDA for ALL in 2026, with the aim of securing product approval by 2027.

In addition to their recent achievements, in the year 2020, Curocell took a momentous step by establishing Good Manufacturing Practice (GMP) facilities within the Future Medicine Building at Samsung Seoul Hospital. This marked the inauguration of the very first clinical drug production facility of its kind in South Korea. Fast forward to April of this year, they accomplished the construction of the first and largest commercial GMP production facility in the country. Currently, they are in the process of validation for approval, a rigorous self-check for quality verification. It is worth noting that this facility has been constructed to meet global standards, positioning Curocell to not only serve the needs of the South Korean market but also cater to the demands of international markets.
Mr. Kim, in his statement, detailed the composition of their GMP facility, which comprises a total of five units. The first floor of this facility is compartmentalized into five sections, with the initial floor dedicated to the production of viral vectors. The second floor is exclusively dedicated to the production of finished products for CAR-T therapy. Significantly, this facility empowers them to manufacture CAR-T therapies for up to 700 patients annually.
Focusing on ‘Asia Business’ using In-house GMP facilities.
CEO Mr. Kim Geon-su underscored the company's early aspirations as an unlisted entity, stating, "Our primary objective as a non-listed company was to introduce a groundbreaking product, CAR-T therapy, to the South Korean market." He elaborated on their vision, saying, "With our listing, Curocell is poised to embark on a second-stage growth strategy, evolving into a more globally oriented company than our current status."
Curocell has strategic plans to broaden its footprint in the Asian market by leveraging its world-class in-house GMP facility through the "Asia Partnership." Furthermore, the company is actively exploring avenues for generating revenue through contract manufacturing (CMO) and contract development and manufacturing (CDMO) activities, all underpinned by their GMP facility.
The company initiated the application process for a special listing in May, successfully clearing the preliminary listing examination in September. The offering encompasses a total of 1.6 million shares, with a per-share offering price range spanning from $22.04 to $24.77. This offering aims to raise approximately $39.6 million (at the upper end of the offering price range). Consequently, the anticipated market capitalization range falls in the vicinity of KRW $30 million to KRW $33.7 million based on the proposed offering price.
To finalize the offering price, the company intends to initiate demand forecasting for institutional investors from October 20th to 26th, spanning five business days. Following this, general subscriptions are slated to open on October 31st and November 1st, with plans for the company to be listed in mid-November. Leading underwriters for this IPO are Samsung Securities and Mirae Asset Securities.
While concerns have been raised in certain quarters regarding Curocell's valuation, CEO Kim Geon-su addressed these apprehensions by suggesting, "The valuation seems to reflect a comprehensive evaluation of the company's fundamentals and the prevailing market conditions." He also noted the presence of industry experts who believe that, should the company achieve its targeted revenue in the future, the multiple at the upper end of the offering price range may actually be considered relatively low.
관련기사
- "큐로셀, 2025년 CAR-T 치료제 허가 목표… 연 700명 치료제 생산"
- 큐로셀, 증권신고서 제출…11월 코스닥 상장 목표
- 큐로셀, 'CAR-T 치료제 주요 연구자 초청 행사' 개최
- 'CAR-T 치료제' 큐로셀, 코스닥 상장예비심사 통과
- 큐로셀 "성인 급성림프구성백혈병 CAR-T 치료제, 첫 임상환자 투여"
- "큐로셀, 국산 첫 CAR-T 신약 허가 정조준… 연내 상장 목표"
- CAR-T 치료제, 큐로셀 안발셀의 생산기지 대전 공장은 "꿈의 공간"
- 큐로셀, ESMO서 혈액암 분야 '최우수 포스터' 선정
- 큐로셀, CAR-T 치료제 안발셀 임상 2상 완료…"내년 9월 허가 신청"
- 큐로셀 "공모주 사기 유도 웹사이트 주의 요망"
- 바이오텍들이 눈독 들이는 일본... "글로벌 교두보 될까?"

