Spleen-Focused LNP Platform Aims to Cut Cost, Time, and Toxicity in Next-Generation Cell Therapy

Efforts are accelerating to replace high-cost CAR-T therapies—often priced in the millions of dollars—with a single injection. At the center of this shift is in vivo CAR-T, a next-generation approach that generates therapeutic immune cells directly inside the body, bypassing the weeks-long, patient-specific cell manufacturing process required for conventional ex vivo CAR-T.
Over the past year, global pharmaceutical leaders including AstraZeneca, AbbVie, and Gilead have committed billions of dollars through acquisitions and partnerships with in vivo CAR-T developers such as EsoBiotec and Capstan Therapeutics. As of 2025, more than 100 in vivo CAR-T programs are in development worldwide, with early clinical signals—such as complete responses in multiple myeloma—further fueling momentum.
As enthusiasm grows, the field faces a defining technical hurdle: delivery. Achieving precise, efficient targeting at low doses remains critical to realizing in vivo CAR-T’s promise. Against this backdrop, Korean biotech Surginex is entering the race with a spleen-targeted lipid nanoparticle (LNP) platform designed to overcome this bottleneck. HIT News spoke with CEO Se-jun Kim to discuss the company’s strategy and clinical vision.
Ending Treatment Delays that Cost Patients their Lives
Conventional ex vivo CAR-T therapy requires collecting a patient’s immune cells, genetically modifying them outside the body, and reinfusing them weeks later. While highly effective—particularly in hematologic malignancies—the process is costly, complex, and time-intensive.
“With current CAR-T therapies, cell manufacturing and logistics alone can take several weeks, at costs ranging from $350,000 to $1.5 million,” Kim said. “Many patients lose their treatment window as the disease progresses while they wait.”
Drawing on his background as a hepatobiliary and pancreatic surgeon, Kim emphasized the urgency. “For patients with advanced cancer, where every moment counts, month-long delays, aggressive preconditioning, and severe toxicities can directly threaten survival.”
In vivo CAR-T offers a fundamentally different model. As an off-the-shelf injectable therapy, it can be administered immediately after diagnosis, eliminating manufacturing delays and allowing patients to begin treatment without missing the critical therapeutic window.
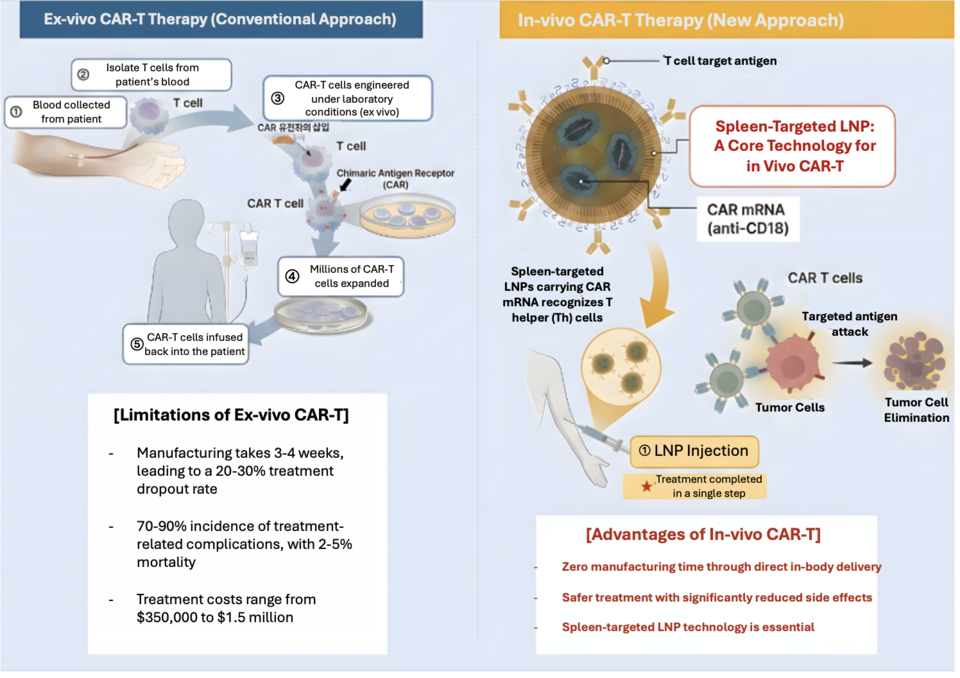
The promise of a “single-injection” CAR-T hinges on one challenge: delivering genetic material precisely to immune cells.
“For in vivo CAR-T to work, LNPs carrying genetic payloads must reach T cells in the body,” Kim said. “The issue is that with conventional LNPs, more than 90% accumulate in the liver.”
Because T cells are concentrated in the blood and lymphoid organs—particularly the spleen—this hepatic sequestration reduces efficacy while increasing toxicity risk.
Surginex’s solution is a spleen-targeted LNP platform. “The key is directing LNPs away from the liver and toward the spleen,” Kim explained. “That’s where immune cells are concentrated.”
The company achieves this through precise physicochemical control of its nanoparticles. By optimizing particle size, surface charge, and lipid composition, Surginex has minimized liver accumulation while significantly improving spleen delivery. This approach both mitigates liver toxicity and enhances therapeutic potency.
Surginex’s lead candidate, RX-171, has demonstrated optimized delivery efficiency in preclinical studies, meeting internal benchmarks based on spleen-to-liver accumulation ratios.
Beyond Cancer: Resetting the Immune System
Surginex’s LNP candidates are rapidly cleared through the liver and kidneys, show low tissue retention, and exhibit favorable safety profiles driven by increased hydrophilicity. These characteristics also make them suitable for repeat dosing—an advantage over viral vectors, which are difficult to administer multiple times.
“Ex vivo CAR-T is typically a one-time treatment,” Kim said. “If those cells become exhausted in the tumor microenvironment, the therapy is effectively over.”
In contrast, in vivo CAR-T allows repeated injections that continuously generate fresh immune cells. “It’s like deploying elite troops again and again until a fortified wall collapses,” Kim said. “That persistence could be decisive against solid tumors, including pancreatic cancer.”
The platform’s flexibility also enables expansion beyond oncology. By keeping the LNP delivery system constant and changing only the encoded mRNA, Surginex can target new diseases. Kim pointed to recent non-human primate studies showing elimination of diseased B cells across blood, spleen, and bone marrow, followed by repopulation with healthy naïve B cells—an effect described as an immune reset.
“This suggests in vivo CAR-T could open a new frontier for refractory autoimmune diseases such as rheumatoid arthritis and lupus,” he said.
Scaling Access Through Low-Cost Manufacturing
From a commercial perspective, Surginex is positioning itself as a delivery-focused platform company. LNPs are well suited to large-scale manufacturing, with processes already validated through COVID-19 mRNA vaccines.
“Gene therapy rests on two pillars: the therapeutic payload and the delivery technology,” Kim said. “Most companies excel at one, not both. That’s why even Moderna and Pfizer rely on externally sourced delivery technologies.”
Surginex is pursuing a B2B licensing model, using a rapid in-house screening system to identify lipid combinations that maximize spleen targeting. Its antibody-conjugation process also mirrors established ADC manufacturing workflows, supporting scalability and cost efficiency.
“Korea has experienced the consequences of lacking proprietary mRNA LNP technology,” Kim said. “With our spleen-targeting platform—an essential enabler of in vivo CAR-T—we aim to bring forward an era where intractable diseases can be treated with just a few injections.”

