Korea leads region with new pembrolizumab indication, offering the first major treatment advance in nearly 20 years

The immuno-oncology drug Keytruda (pembrolizumab) has been approved in Korea as a perioperative adjuvant therapy for head and neck cancer—introducing the first new therapeutic option in almost 20 years.
MSD Korea held a media seminar on the 2nd to discuss the October approval, which marks the first such indication granted in the Asia-Pacific region.
Professor Hye-ryun Kim, medical oncologist at the Yonsei Cancer Center, explained that head and neck cancer arises in areas such as the tongue, lips, retromolar region, salivary glands, larynx, and nasopharynx. Because these areas are essential for speech and eating, patients often face severe functional impairment.

Professor Kim noted that head and neck cancer is the sixth most common cancer globally. The worldwide patient population is expected to increase from 947,211 cases in 2022 to 1,452,718 cases by 2045, with approximately 45,000 patients currently estimated in Korea.
Diagnosis is frequently delayed—50.2% of cases are detected at stage III–IV—resulting in a 5-year survival rate of only 20–30%. Even when tumors are surgically resectable, survival remains around 50%, underscoring the urgent need for improved treatment strategies.
Compared with cancers such as lung or breast cancer, head and neck cancer has historically had fewer effective treatment options. Existing therapies have delivered limited clinical benefit, highlighting a significant unmet need. Keytruda, a PD-1 inhibitor that restores T-cell activity by blocking the PD-1/PD-L1 pathway, is therefore viewed as a meaningful addition to the treatment landscape, particularly for recurrence prevention.
Professor Kim presented updated data from KEYNOTE-048, a global phase 3 study evaluating Keytruda monotherapy and Keytruda plus chemotherapy in recurrent or metastatic patients who had not received prior systemic therapy.
Keytruda monotherapy achieved a median overall survival (mOS) of 11.5 months, while the Keytruda + chemotherapy arm reached 13 months, both exceeding the 10.7 months seen with standard treatment. Five-year survival reached 15% with Keytruda monotherapy—more than double the 6% achieved with conventional therapy.
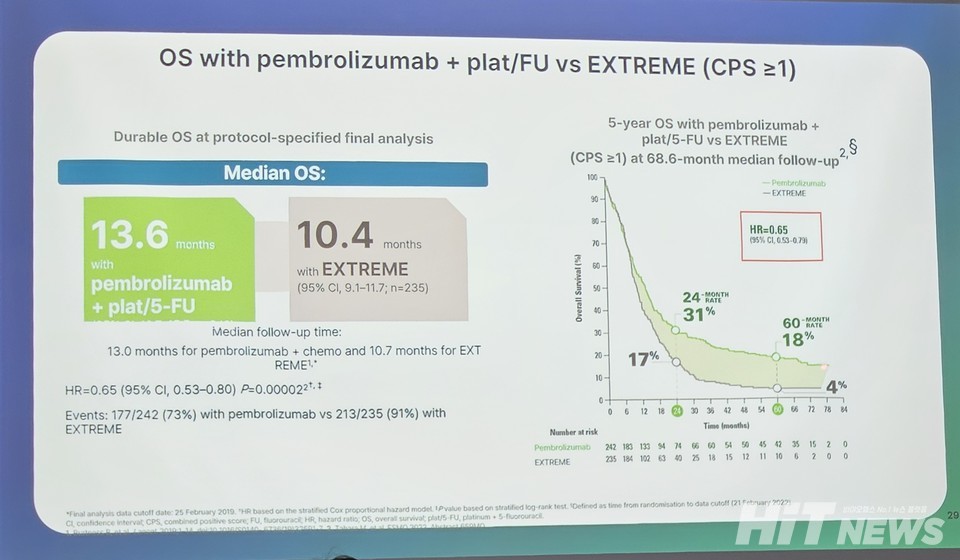
Among patients with PD-L1 CPS ≥20, mOS was 14.8 months with Keytruda monotherapy, 14.7 months with Keytruda plus chemotherapy, and 11 months with standard therapy.
KEYNOTE-689, which evaluated two cycles of preoperative Keytruda in resectable stage III–IV patients, showed a 30% reduction in recurrence risk compared with standard therapy. Overall survival also improved significantly—68.2% with Keytruda vs 59.2% with conventional treatment.
“Keytruda is now recognized as a standard therapy for head and neck cancer in the ASCO guidelines,” Professor Kim emphasized. “Above all, multidisciplinary care remains essential.”

Professor Hyun-joon Hong, otolaryngology specialist at the Yonsei Cancer Center, highlighted the unique burden of head and neck cancer. “Lung, liver, and stomach cancers are also serious, but they often progress without visible signs,” he said. “In contrast, head and neck cancer can visibly alter the face and impair breathing.”
He explained that lymph node metastasis is common and significantly worsens prognosis. Treatment also varies widely depending on tumor location and individual patient characteristics, making management highly complex. Because the disease affects breathing, eating, and speaking, patients frequently experience challenges in daily functioning and social interaction.
“Korea sees about 5,000 new cases of head and neck squamous cell carcinoma each year,” Professor Hong said. “A tumor the size of a 100-won coin corresponds to stage I; the size of a 500-won coin corresponds to stage II. Spread to adjacent tissues indicates stage III, and metastasis to the lungs or bones indicates stage IV.”
He added, “There is no single correct treatment approach, which is why gathering robust clinical data is essential. Keytruda is expected to contribute meaningfully to this effort. Improving survival for head and neck cancer patients requires close collaboration among patients, caregivers, and healthcare professionals through a multidisciplinary approach.”

