Dose-based pricing strategy and distributor engagement signal market readiness amid intensifying competition with Wegovy
Korea Lilly is expected to launch its anti-obesity drug Zepbound—positioned as a direct competitor to Novo Nordisk’s Wegovy—as early as late August. Industry sources confirm that the company has already held detailed discussions with major pharmaceutical distributors regarding pricing and distribution plans.
According to those familiar with the matter, Korea Lilly recently met with over 30 South Korean distributors to outline its launch strategy and pricing approach. While the final ex-factory price has not been confirmed, it is reportedly being considered at a level slightly below that of Wegovy.
A key differentiator lies in Lilly’s pricing strategy: unlike Wegovy’s flat-rate pricing across all dosages, Zepbound will follow a tiered model based on dose strength. Distributors were also informed of the anticipated timeline, with late August being cited as the target launch window, although no official date has been announced.
The concrete nature of these meetings—beyond informal rumors—signals significant progress toward market entry. Industry observers note that while pre-launch briefings with clinicians are standard, engagement with distributors and contract sales organizations (CSOs) often reflects a more advanced stage of preparation.
Initial discussions about Zepbound’s launch began in 2024. Although there was talk of potential co-promotion with another company in late 2023 and early 2024, Lilly ultimately opted for an independent sales model. The launch, originally planned for the first half of 2025, was delayed due to internal preparations.
In the meantime, rival Novo Nordisk strengthened its market position. The company initiated reimbursement efforts for its diabetes drug Ozempic in May and resolved early supply issues with Wegovy, leading to a significant sales uptick—adding competitive pressure on Lilly.
Pricing considerations are also tied to broader reimbursement efforts. In April, the Korean Diabetes Association revealed during a press conference that it is collaborating with pharmaceutical companies to pursue reimbursement for GLP-1 therapies. Medical sources indicate that the association held discussions in May with GLP-1 drug manufacturers, further supporting the recent developments regarding Zepbound’s pricing and timing.
Notably, Lilly’s adoption of dose-based pricing could reflect strategic positioning for future reimbursement eligibility. While Wegovy remains a non-reimbursed obesity therapy with uniform pricing, Zepbound’s structure may allow greater pricing flexibility—particularly if future indications for diabetes treatment are sought.
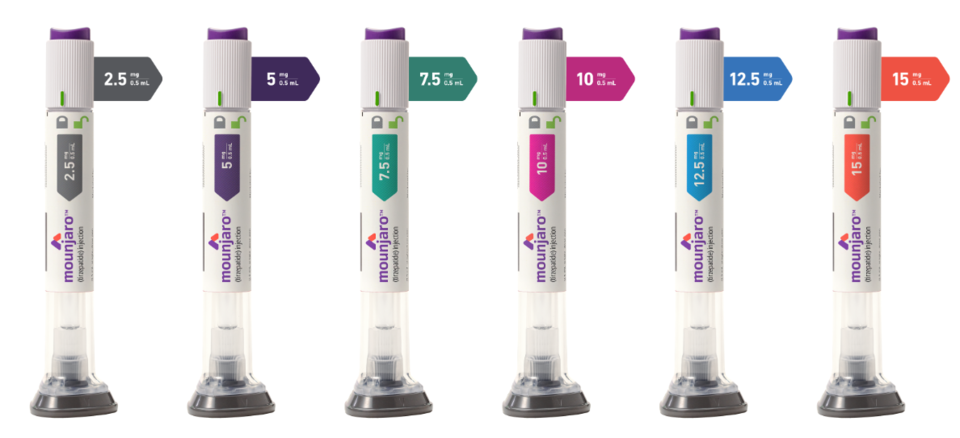
Zepbound is expected to be offered in six dosage strengths: 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, and 15 mg—outpacing Wegovy’s five-tier system, which ranges from 0.25 mg to 2.4 mg. Industry stakeholders note that Wegovy’s higher-dose prescriptions have led to “split-dosing” workarounds, causing supply issues and distributor dissatisfaction. Zepbound’s expanded range may help preempt similar challenges.
When asked about pricing and launch details, a Korea Lilly representative stated, “The price and launch date have not yet been finalized.”

