Tighter Focus on High-Cost Drugs, Expanded Indications, and Cost-Effectiveness

The National Health Insurance Service (NHIS) plans to strengthen oversight of pharmaceutical expenditures by enhancing post-pricing management systems, including Price-Volume Agreements (PVA) and negotiations for expanded drug indications.
At a press briefing on April 15, Yoo-kyung Yoon, Director of the Pharmaceutical Benefits Management Division, shared the agency’s 2024 agenda.
In 2023, drug spending reached $18.5 billion, accounting for 23.6% of the national health insurance budget, up 0.8 percentage points from the previous year. This marks a continued rise from $14 billion in 2019.
To address this trend, NHIS conducted a foundational study last year to broaden financial analysis related to PVA and initiated reforms to improve cost-efficiency.
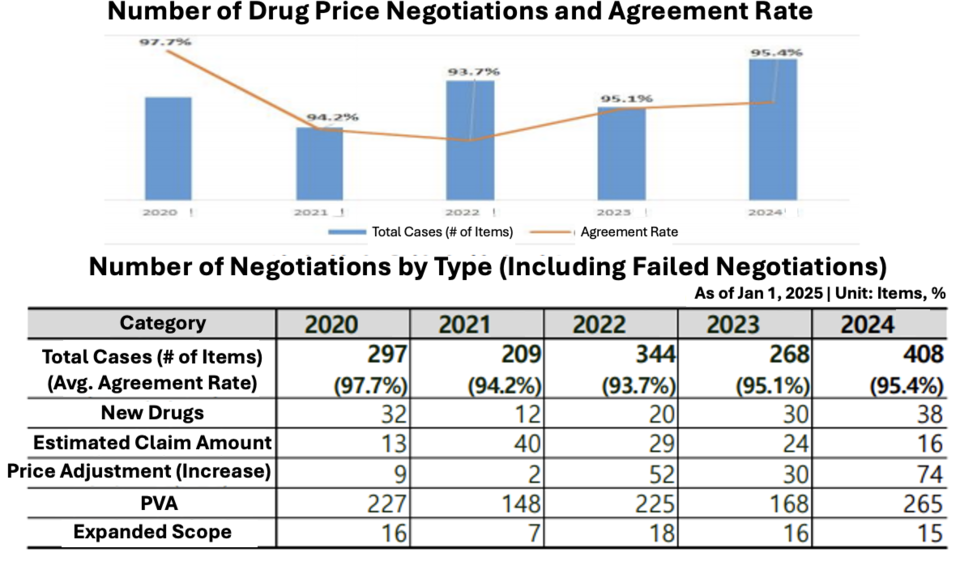
Yoon reported that the NHIS applied differentiated pricing based on reimbursement volume under the PVA system, raising discount rates for high-cost drugs by 36% and easing exemption standards for smaller pharmaceutical firms. These negotiations, covering 257 items, resulted in $66 million in savings.
For 2024, NHIS will prioritize:
★ Managing reimbursement expansion for high-cost drugs
★ Monitoring and negotiating clawbacks for drugs under clinical re-evaluation
★ Refining the PVA framework
★ Conducting indication expansion negotiations
Under the updated PVA system, NHIS will enforce a maximum price reduction of 12.5% for high-cost drugs to enhance expenditure control.
Negotiations for expanded indications will focus on therapies such as immuno-oncology drugs. These will involve international benchmarking, cost-effectiveness assessments, budget impact reviews, and post-listing evaluations—reflecting increased demand for broader coverage of costly cancer and one-time curative treatments.
To support these initiatives, NHIS is forming a consultative group with pharmaceutical companies to develop detailed guidelines that incorporate financial impact analyses and drug-specific factors. The goal is to balance fiscal sustainability with public health benefits.
Yoon also addressed the ongoing lawsuit over the clawback nullification for choline alfoscerate. The agency is working with the Ministry of Health and Welfare on strategies, including monitoring withdrawn approvals and developing legal countermeasures.
Hyun-deok Kim, Head of the Drug Pricing Reform Division, added, “Many of choline’s indications have been delisted, and 25% of prescriptions are for unapproved uses. We will continue to monitor clawback negotiations and seek ways to inform the public about off-label prescribing.”
Se-rim Oh, Head of the Post-Negotiation Management Division, emphasized the importance of identifying whether companies exiting mid-process do so due to failed re-evaluations or poor sales performance. “We are reviewing this from both legal and operational angles to ensure a sound response,” she said.

