Merger with EPD Biotherapeutics Drives R&D Synergy and Next-Generation Growth
"We are committed to developing new drugs that can overcome the limitations of traditional PROTACs by utilizing the 'bioPROTAC' platform, which holds potential to aid patient treatments. Our goal is to achieve global licensing-out (L/O) of our bioPROTAC-based pipeline, 'GX-BP1 (SOX2 bioPROTAC),' by the first half of 2026," said Jae-hyun Choi, Executive Vice President and Head of Research and Development (R&D) at Genexine, during a recent interview with Hit News.
A seasoned expert in oncology drug development, Choi oversees all R&D-related functions at Genexine, including bio-research and clinical development. Genexine, founded in 1999 as a first-generation South Korean biotech company, has built expertise in discovery, manufacturing (CMC), and clinical development. In October, the company completed a merger with EPD Biotherapeutics, a targeted protein degradation (TPD) developer, acquiring the proprietary bioPROTAC platform technology, 'EPDeg,' along with key research personnel.
Choi, who previously served as CEO of EpD Bio and contributed to PROTAC development at Arvinas, shared his vision for advancing Genexine's bioPROTAC pipeline and the company’s broader growth strategy.

Merger with EPD Bio Brings BioPROTAC Platform and R&D Talent
Explaining the merger, Choi said, "As an early-stage biotech, EPD Bio required stable funding, while Genexine sought partners to secure talent and assets for next-generation growth opportunities. The merger aligned these mutual needs."
He emphasized that the collaboration integrates EPD Bio’s bioPROTAC platform with Genexine’s clinical development expertise. "This synergy combines Genexine's strengths in protein engineering, CMC expertise, and clinical development with EpD Bio's early-stage innovation," Choi added.
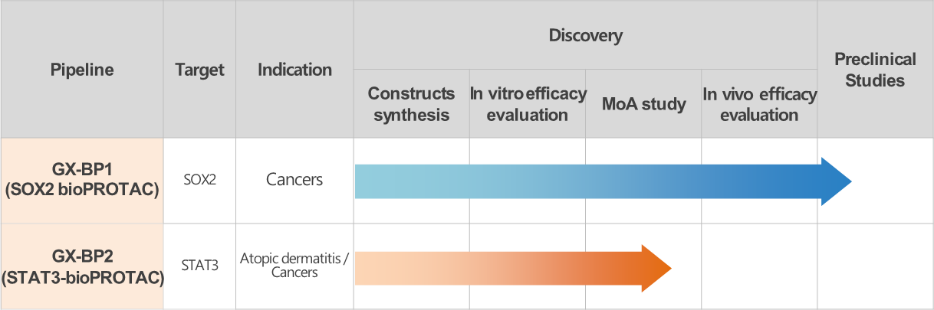
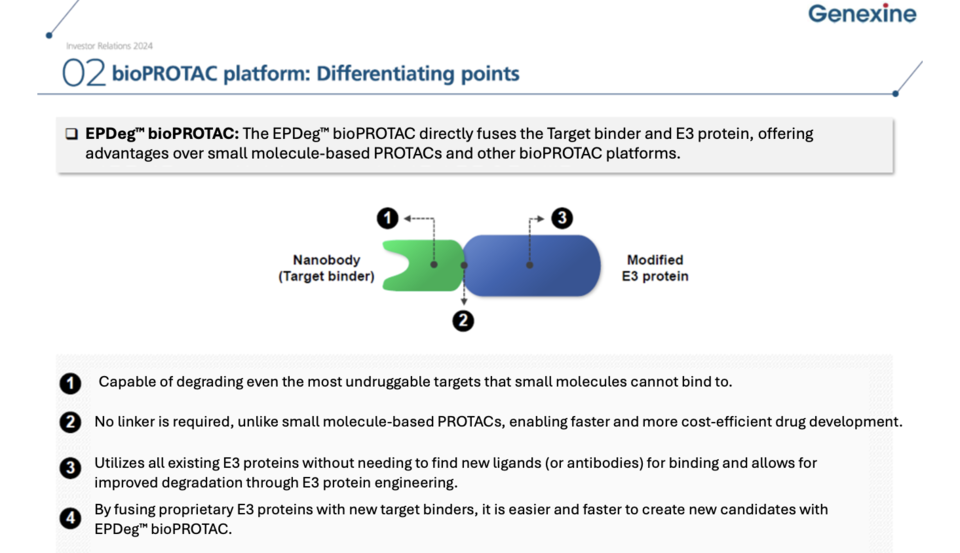
Through the merger, Genexine incorporated 15 EPD Bio personnel, including 14 R&D professionals. Currently, Genexine's bioPROTAC-based pipeline includes GX-BP1 (SOX2 bioPROTAC) and GX-BP2 (STAT3 bioPROTAC). GX-BP1, targeting the SOX2 protein, is in preclinical development and has shown effective protein degradation and anticancer efficacy in vivo. It is being developed for non-small cell lung cancer, while GX-BP2, targeting STAT3, is being developed for atopic dermatitis.
SOX2, a key oncogenic transcription factor, plays a critical role in cancer stem cell stemness, growth, metastasis, and immune evasion. It is highly expressed in lung squamous cell carcinoma, a subtype of non-small cell lung cancer.
Takeda Collaboration for GX-BP1 Research Highlights Global Interest

Choi noted, "Over the past decade, more than 80% of global deals in the TPD field occurred at the discovery stage. Genexine plans to focus on licensing-out its early-stage bioPROTAC pipelines."
Currently, GX-BP1 is being developed through a joint research project funded by Takeda Pharmaceuticals, which is interested in addressing the unmet medical needs of lung squamous cell carcinoma. "We aim to license out GX-BP1 during its preclinical stage," Choi added.
Genexine also plans to commercialize its existing pipeline, such as the anemia treatment Epeesa, while driving next-generation growth through the bioPROTAC platform.
Concluding the interview, Choi outlined plans for open innovation, stating, "Genexine intends to collaborate with companies possessing drug delivery system (DDS) technologies essential for mRNA therapeutics. By adopting an agile R&D strategy, we aim to maintain our competitiveness in the global biotech landscape."

