Supporting Local Production in West and Central Africa, 30 Million Doses Annually
June Agreement with South Africa's Biovac for Pentavalent Meningococcal Vaccine
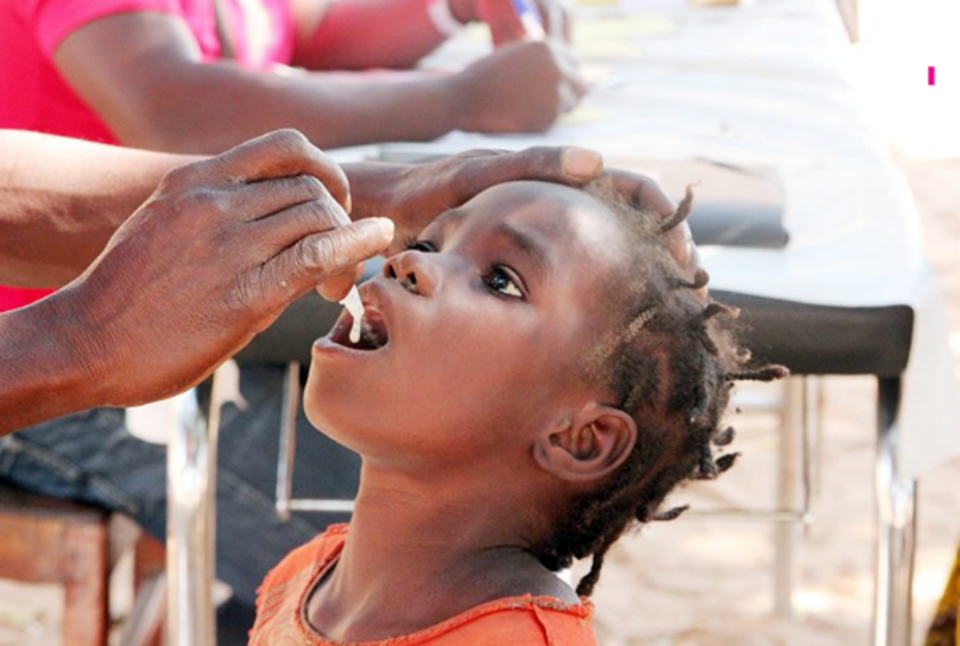
Eu Biologics signs agreements with African vaccine manufacturers for the supply of oral cholera vaccine (OCV) and meningococcal vaccine bulk, along with the transfer of finished product manufacturing technology. This strategic move is expected to expedite its entry into the African market.
On August 4th, industry sources reported that Eu Biologics (CEO Young-ok Baek) entered into an agreement on July 31 with DEK Vaccines Ltd, a vaccine manufacturer in Ghana, West Africa, for technology transfer and bulk supply for the production of oral cholera vaccine. This agreement follows a memorandum of understanding (MOU) signed in September last year. Through this contract, DEK will be able to produce the finished product of Eu Biologics' oral cholera vaccine, 'Euvichol-S,' directly in Africa. DEK expects to launch Euvichol-S by the end of 2026 after completing the technology transfer and approval process.
Eu Biologics plans to support local production in West and Central African countries, with a maximum annual supply limited to 30 million doses. A company representative stated, "Recent simultaneous outbreaks of cholera in East Africa (Kenya, Ethiopia), Southern Africa (Zambia, Zimbabwe), and West Africa (Nigeria, Cameroon) highlight the need for oral cholera vaccine supply."
The representative further explained, "DEK is capable of entering the market with plastic tube-type vaccines. DEK's parent company, Kinapharma, possesses a sales network in West and Central Africa, which is expected to be leveraged. The company anticipates demand from public markets, including UNICEF and the health ministries of West and Central African countries."
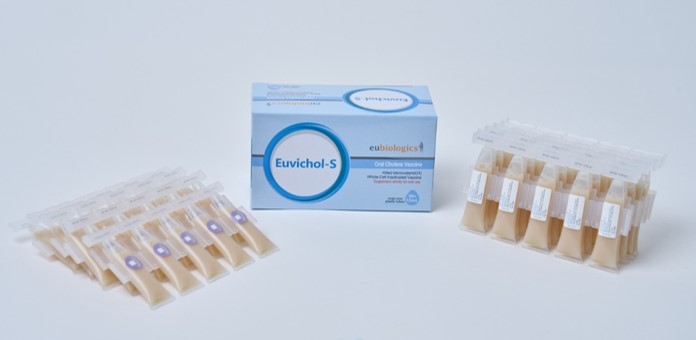
A representative from Eu Biologics stated in a press release, "The contract for DEK's technology transfer and bulk supply of the oral cholera vaccine signifies the diversification of cholera vaccine sales, which have been concentrated on UNICEF. Following the cholera vaccine, we will also support local production of typhoid and meningococcal vaccines. By expanding bulk sales of public vaccines, we anticipate stable sales growth and increased profitability."
Previously, in June, the company signed a technology transfer agreement with Biovac, a South African vaccine manufacturer, for the pentavalent meningococcal vaccine. Biovac is a major vaccine company in South Africa, with a vaccine pipeline that includes tuberculosis, measles, pneumococcal, and hepatitis B vaccines, and supplies vaccines and other biologicals to Africa and neighboring regions.
Eu Biologics expects the technology transfer agreement with Biovac to bridge gaps in the current prevention and treatment of meningitis in sub-Saharan Africa, particularly in the meningitis belt stretching from western Senegal to eastern Ethiopia, where the unique serotype X circulates.
The representative further emphasized, "Africa is a key market for vaccines such as cholera, typhoid, and meningococcal vaccines. Eu Biologics will collaborate with large local pharmaceutical companies in Africa, as well as with UNICEF, the World Health Organization (WHO), and the Global Alliance for Vaccines and Immunization (GAVI) to contribute to improving health in Africa."

