Boryung’s Triumph: Market Leadership, Global Manufacturing, and Innovative Anticancer Development
In 2024, Hit News is set to collaborate once again with the Korea Pharmaceutical Bio Manufacturers Association (KPBMA)'s specialized press corps for exclusive interviews with CEOs of major pharmaceutical companies. The interviews will follow a 'Year to Year' format, reflecting on the accomplishments of 2023 and outlining plans for the new year. The exclusive interview with Boryung's CEO Doo-Hyun Jang took place at a restaurant in Seoul's Gwanghwamun on February 5th. The attention-grabbing title of the recently distributed interview material with Boryung, "Advancing the Sovereignty of Anticancer Drugs," set the tone for the discussion.
① The strategy we refer to as LBA (Legacy Brands Acquisition) has been pivotal for us. In 2020, we acquired the anticancer drug Gemzar, followed by the schizophrenia treatment Zyprexa in 2021, and the non-small cell lung cancer treatment Alimta in 2022. Our focus lies in acquiring products with a clear market and guaranteed revenue, significantly aiding our company's data-driven management. We aim to sustain M&A activities targeting game-changer products capable of yielding even greater profits.
② Pursuing this path is imperative for Boryung. In the next 3-5 years, we must compete with large pharmaceutical companies in terms of scale, a challenge that cannot be met through the same approach. Our objective is to build a portfolio allowing us to sell not just one but multiple products, engage in discussions with experts, and broaden our horizons. Whether through licensing, M&A, or the development of generic or improved drugs, creating a comprehensive portfolio is my role.
③ Our primary focus is on diabetes. Securing a foothold in the diabetes market stands as one of our management objectives. This year alone, patents for drugs like Farxiga and Januvia will expire. We have prepared 15 diabetes-related products, including single and combination drugs, set to launch in the next five years. With a market opening of $1.5 to 2.2 billion, capturing just 10% translates to $150 million.

Boryung has preliminary reported sales of $643 million and an operating profit of $51 million based on the consolidated financial statements for the year 2023. When compared to the performance sheets of 2022 ($569 million - $42 million) and 2021 ($469 million - $31 million), this indicates an annual external growth of over $75 million and a profit growth of over $7.5 million. Considering the industry's historical performance, one could describe this as an 'acceleration' that is not without merit. CEO Jang's assurance of the success of the LBA strategy and dominance in the diabetes generic market during the early 2023 press meeting has materialized, yielding returns in cash on the expected date.
In contrast to his previous interview style, CEO Doo-Hyun Jang has adopted a more composed demeanor, stating, "We are aiming for sales exceeding $748 million and operating profit surpassing $748 million." Designating 2024 as the "inaugural year" for becoming the industry's number one, he is actively propelling Boryung forward with the number 1 as the focal point. Expressing his ambition, he envisions this year as the starting point for preparing and aligning working methods, systems, and more in line with the standards of the industry leader.
CEO Jang emphasized Boryung’s commitment to reaffirming its business and distribution capabilities, aiming for a year of drive and verification. In addition to the joint sale of K-CAB with HK inno.N, he highlighted continuity in the portfolio. Key personnel in anticancer operations and R&D, including Bong-Seok Kim, Ki-Deok Kim, and Young-Seok Kim, were present at the meeting. Jang urged interest in Boryung’s positioning and leadership in Oncology.

Anticancer Business Keyword ① South Korean Market Share No. 1
Leveraging its competitive strength in anticancer drugs, Boryung has achieved a significant business milestone by becoming the leading pharmaceutical company in market share within South Korea. The scale of prescriptions for anticancer drugs in South Korea is approximately $2.2 billion (based on IQVIA data, 2022.4Q~2023.3Q), with a substantial dependence on foreign products, constituting 76.4% of the market. Among the top 10 companies in prescription volume, Boryung stands out as the sole Korean company, securing the fourth position with sales reaching $185 million. Currently, Boryung offers a diverse portfolio of over 30 products, including high-cost items such as Boryung Epirubicin HCl Injection (Epirubicin), EPS Injection (Etoposide), and A.D. Mycin Injection (Doxorubicin), with a cost ratio exceeding 100%.
Boryung not only concentrates on the development and stable supply of K-anticancer drugs but also directs its research and development efforts towards enhancing the convenience of existing products to improve conditions for cancer treatment. In the past year, Boryung achieved a notable enhancement by transforming the formulation of the anticancer drug Gemzar (active ingredient Gemcitabine) from the traditional powder form to a liquid injection. Furthermore, the company introduced the first South Korean-produced alcohol-free docetaxel liquid formulation anticancer drug, 'Ditaxel.'
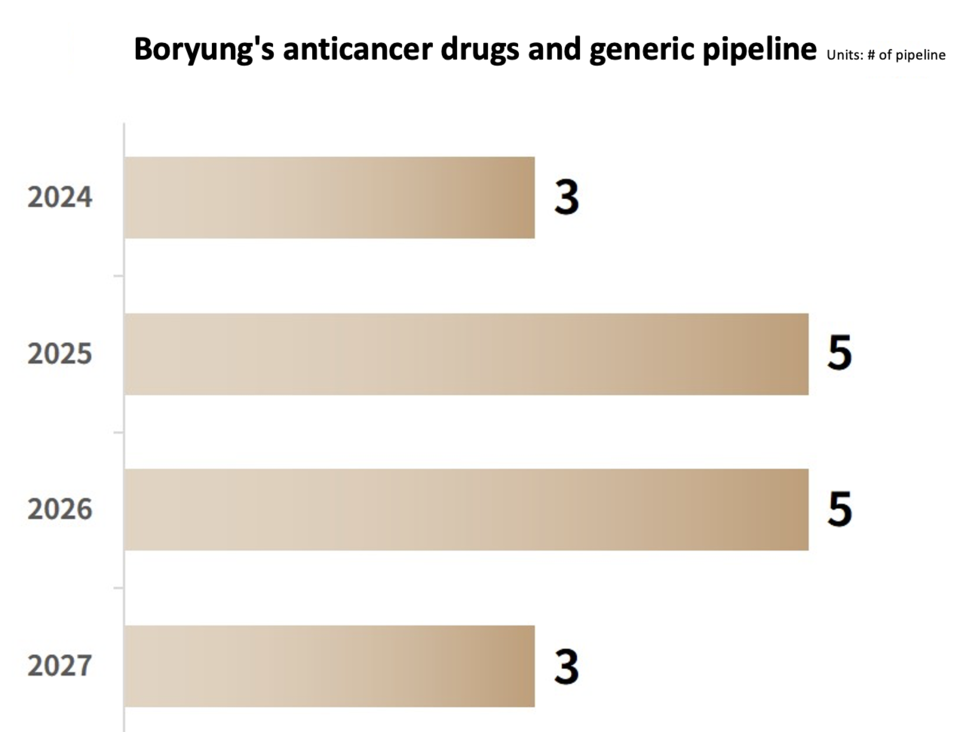
Anticancer Business Keyword ② 4-Fold Growth with Specialized Terms and LBA Strategy
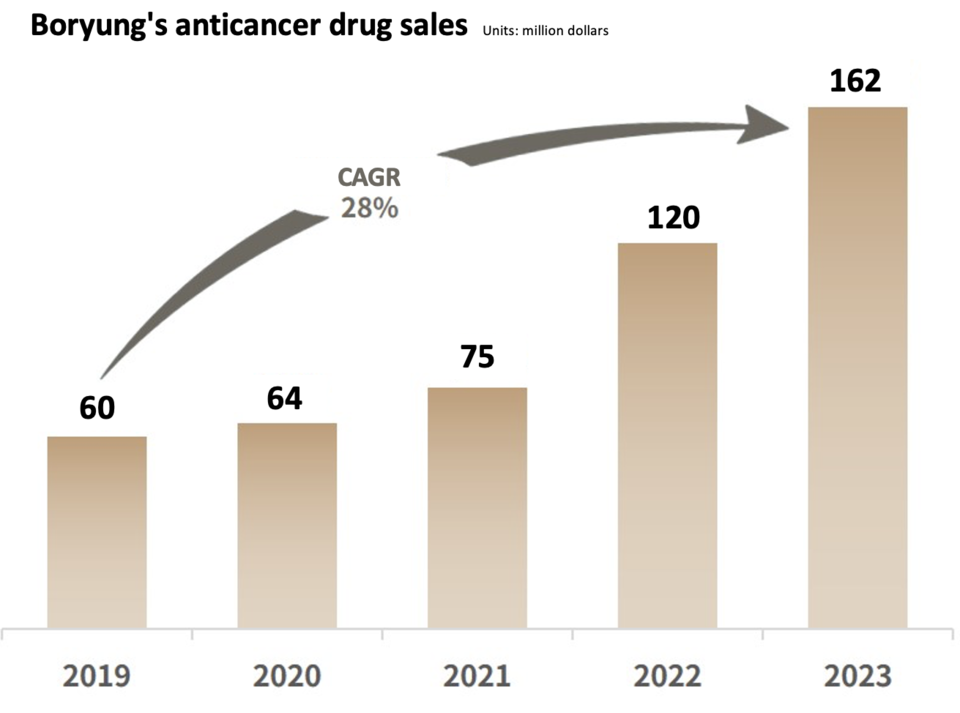
Since 2007, Boryung has operated an 'Anticancer Drug Specialized Team,' taking a strategic step in 2019 to establish the 'Onco Division,' which further expanded into the 'Onco Department' in 2020. In 2021, the company pioneered the formation of the only specialized group for hematologic cancers in Korea, and as of January this year, a dedicated lung cancer team has been established. This deliberate structure ensures a specialized approach with distinct knowledge and experience for each type of cancer. Building on this foundation, Boryung's anticancer drug business has witnessed an impressive nearly threefold growth, escalating from sales of $59.7 million in 2019 to $162 million in 2023, achieved within just four years.
Remarkably, Boryung, through its distinctive 'Legacy Brands Acquisition (LBA) Strategy,' has successfully commercialized and internalized globally prevalent anticancer drugs. This strategy guarantees a stable supply while concurrently accelerating sales and profitability. The LBA strategy entails acquiring original pharmaceuticals capable of maintaining a high market share even after patent expiration. Boryung has implemented this strategy by acquiring global anticancer drugs such as Gemzar and Alimta from international pharmaceutical companies, showcasing its dedication to sustained growth in the field.
Anticancer Business Keyword ③ EU GMP Approved Budget Anticancer Drug Facility
Boryung boasts a world-class facility and a team of expert personnel capable of large-scale production of anticancer drugs, ensuring a consistent and reliable supply. The anticancer injectable drug production facility, completed in 2019, features a cutting-edge 'Isolator System' integrated into most manufacturing processes, ensuring the safe production of highly potent pharmaceuticals. The Isolator acts as a protective barrier, preventing direct exposure of operators to hazardous substances.
In November 2020, following Good Manufacturing Practice (GMP) approval from the Ministry of Food and Drug Safety (MFDS) in South Korea, Boryung initiated the production of the multiple myeloma treatment 'Velkin (active ingredient Bortezomib)' at the budget facility, concluding by the end of the same year. Notably, in February, the facility also secured EU-GMP (European Union Good Manufacturing Practice) certification. Boryung showcased its ability to directly manufacture globally acquired anticancer drugs, such as Gemzar, at the budget campus, underscoring its proficiency in global anticancer drug manufacturing through its LBA strategy.

Anticancer Business Keyword ④ Rare Cancer Peripheral T-Cell Lymphoma New Drug Development
Boryung is presently engaged in the development of an innovative anticancer drug, 'BR101801 (Project Name BR2002),' specifically designed for rare cancers. BR101801 represents a groundbreaking candidate substance that concurrently inhibits key growth regulators in cancer cells, targeting PI3K gamma (γ), PI3K delta (δ), and DNA-PK. This drug is being developed as a treatment for Peripheral T-Cell Lymphoma (PTCL). The recent completion of Phase 1b clinical trials, involving a total of 19 patients (resulting in 2 complete responses and 1 partial response), along with the completion of Phase 1a clinical trials in 2021 (yielding 1 complete response and 2 partial responses), has affirmed its efficacy in six patients. These clinical findings were presented at the American Society of Hematology (ASH), the world's largest hematology conference.
BR101801 received orphan drug designation from the U.S. FDA in October 2022 and rare disease designation from the MFDS in South Korea in August 2023. Boryung plans to finalize the Phase 1 results report in the first quarter of this year and aims to submit an application for Phase 2 clinical trials within the same year.
Moving forward, Boryung envisions concentrating on the NRDO (No Research Development Only) strategy for drug development. NRDO involves rapidly advancing into the clinical stage by introducing drug candidates from external sources such as biotech. To realize this strategy, Boryung is actively pursuing collaborative research projects with promising biotech companies.


