
Daewoong Pharmaceuticals achieves a breakthrough at its Osong plant, securing 'Data Integrity (DI)' and bolstering cost competitiveness through a specialized GMP-focused IT solution from its subsidiary, IDS&TRUST. The plant's recognition with a 4th-stage Smart Factory certification by the Korea Technology and Information Promotion Agency for SMEs (TIPA) highlights its proactive approach and advanced IT solutions, enabling process operation simulations.
In an exclusive interview conducted by HITNews in collaboration with the Korea Pharmaceutical and Bio-Pharma Association (KPBMA), Seung-Ha Lee, Director of the Osong plant, and Jung-Jae Lee, Head of IDS&TRUST, shed light on the significance of the Smart Factory certification and the adoption of GMP solutions.

Three elements of a smart factory: automation, unmanned operation, and digitization
Director Seung-Ha Lee articulated the strategic adoption of a smart factory, citing the Fourth Industrial Revolution and escalating pharmaceutical quality regulations as primary drivers. The $162 million investment made in 2017 aligned with cGMP standards of the US FDA, focusing on automation, unmanned operations, and digitization to fortify the plant's capabilities.
The implementation of IT solutions aimed at preventing errors in manufacturing processes, coupled with a tamper-proof data management system and real-time monitoring, underscores the plant's pursuit of superior quality and cost competitiveness globally.
Moreover, Director Lee highlighted the unique challenges in pharmaceutical smart factories, emphasizing the critical need for stringent process management to uphold quality standards, especially in raw material handling, manufacturing, and distribution.
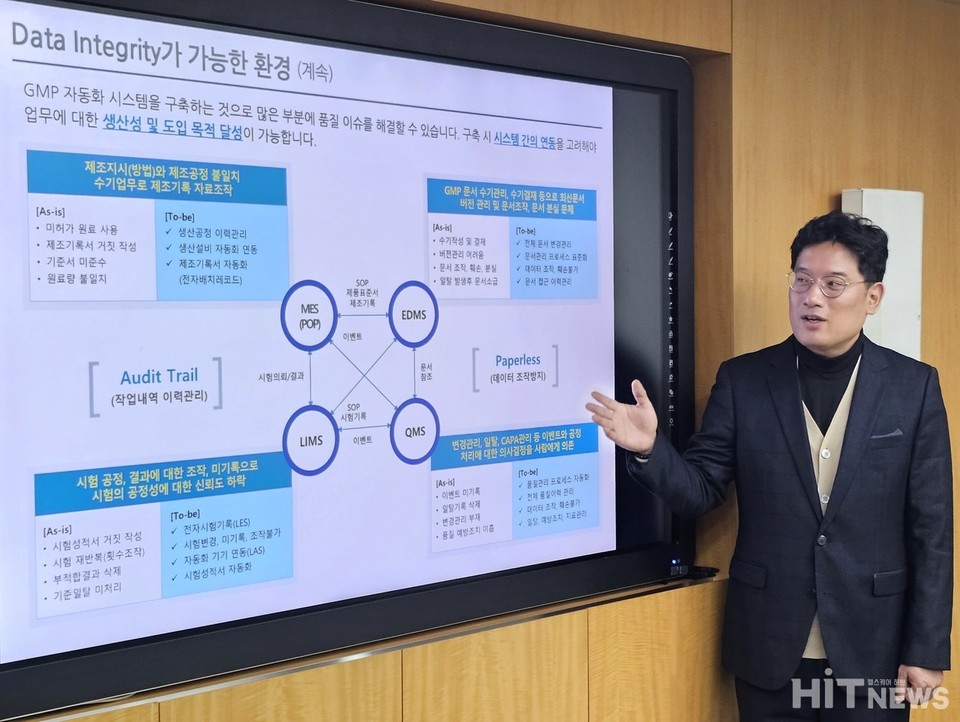
Empowering Quality: implementing IDS&TRUST’s GMP specialized solution

The integration of the 'GxP Solution' from IDS&TRUST automates and records processes within the GMP system, ensuring compliance with industry regulations. This integration spans MES, LIMS, EDMS, and QMS, enabling global GMP compliance, automated operations, continuous monitoring, and substantial improvements in productivity and cost efficiency.
Real-time monitoring and a 'paperless' factory concept are central to operations, where data records and logs are automatically managed through a customized solution, ensuring efficient operations and creating new value through digitally transformed data.
The Osong plant's success story extends beyond quality management, demonstrating a remarkable 40% improvement in productivity and over 30% reduction in costs, setting it apart from similar-sized facilities. The emphasis on staff training and their engagement with cutting-edge technologies like big data, machine learning, and AI further drives operational excellence.
Director Lee accentuated the necessity of bold investment and specialized talent in operating a smart factory, crediting proactive leadership and support organizations like the 'AI Propulsion Team' and the 'Smart Factory Department' for the plant's achievements.
Looking ahead, the vision is to elevate the Osong plant to a Stage 5 smart factory, leveraging national initiatives on AI/machine learning and continuing system upgrades and talent development to set new standards in pharmaceutical production.

