Remibrutinib offers potential alternative to injectables, showing strong Phase 3 efficacy in antihistamine-refractory patients
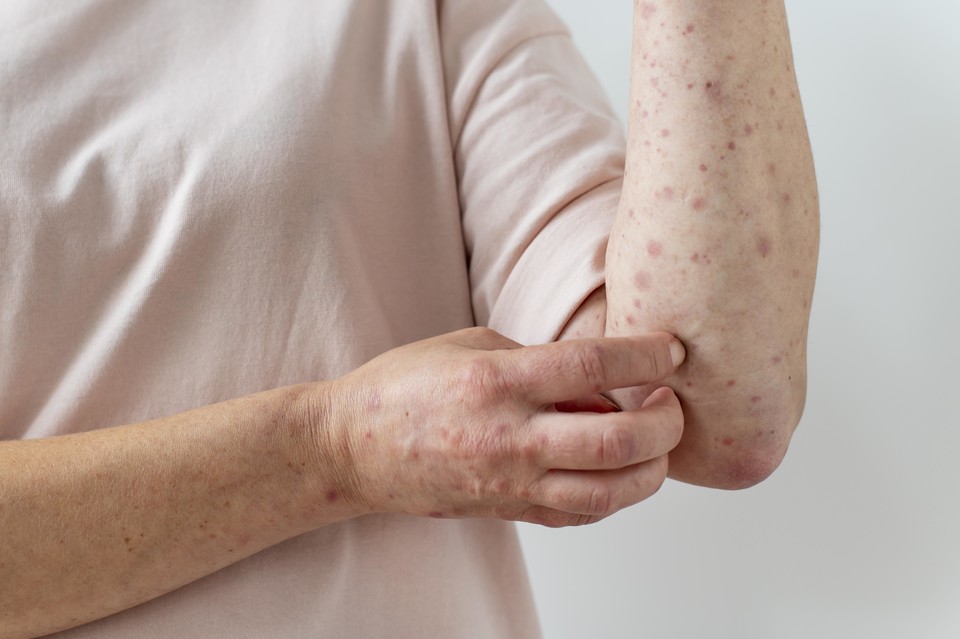
Novartis Korea has filed for South Korean approval of its oral chronic spontaneous urticaria (CSU) treatment, Rhapsido (remibrutinib), positioning it as a potential new therapeutic option.
According to industry sources on March 20, the company submitted a marketing authorization application to the Ministry of Food and Drug Safety (MFDS), expanding its portfolio beyond the injectable Xolair (omalizumab) to include an oral therapy.
CSU is characterized by recurrent wheals or skin redness lasting more than six weeks without a known cause. First-line treatment typically involves third-generation antihistamines, with dose escalation or immunosuppressants used in refractory cases. However, long-term use of these treatments can lead to side effects, including fatigue, cognitive impairment, gastrointestinal issues, and reduced immunity.
Rhapsido, an oral Bruton’s tyrosine kinase (BTK) inhibitor, received U.S. FDA approval in September last year. In Phase 3 REMIX-1 and REMIX-2 trials, it demonstrated significant improvements over placebo at week 12 in itch severity, hives, and overall disease activity among patients inadequately controlled on antihistamines.
As an oral option, the drug may provide an alternative for patients hesitant to switch to injectable therapies.
Meanwhile, Novartis Korea has launched the “Mandu’s Dream” awareness campaign via its official Instagram channel. The campaign features 12 short-form videos over three weeks, focusing on diagnosis, daily challenges, and patient communication, alongside planned collaborations with influencers to raise public awareness of CSU.

