MSD Korea adds 11 new reimbursed indications, easing treatment burden for patients with gastrointestinal and women’s cancers

Pembrolizumab (Keytruda) has expanded its reimbursement coverage in South Korea following a three-year effort, increasing the number of reimbursed indications from seven to 18. The expansion is expected to significantly reduce the financial burden for patients with gastrointestinal and gynecologic cancers, who previously had limited treatment options.
On January 29, MSD Korea held a press briefing to announce the change. As of February 1, reimbursement applies to 11 additional indications, including gastric cancer, esophageal cancer, and triple-negative breast cancer.
At the event, Professor Geun-wook Lee of the Division of Hematology and Oncology at Bundang Seoul National University Hospital stated, “Fifteen years after conventional therapies entered the reimbursement system in Korea, Keytruda has now been incorporated as a reimbursed immuno-oncology combination option.”

Professor Lee noted that gastric cancer ranks fifth globally in both incidence and mortality, with approximately 30,000 new cases diagnosed annually in Korea. Despite improved screening and early detection, around 30% of patients are still diagnosed at a locally advanced or metastatic stage. The five-year survival rate for metastatic gastric cancer remains below 10%, lower than that of lung cancer at approximately 12.1%.
For HER2-positive gastric cancer, the previous standard regimen of trastuzumab plus chemotherapy achieved a median overall survival (mOS) of 16 months, compared with 11.8 months in the placebo group. Subsequent HER2-targeted trials failed to demonstrate benefit, leaving a treatment gap for more than a decade.
With Keytruda, mOS increased to 20 months, representing a 3.2-month improvement over the control group (16.8 months). In patients with PD-L1 CPS ≥1, median survival exceeded 20 months, and the objective response rate was higher with pembrolizumab (72.6%) than with placebo (60.1%).
“Fifteen years after the last reimbursed option for HER2-positive disease, a new standard of care has emerged,” Professor Lee said, noting that a complete response rate of 17% now allows patients to realistically hope for long-term survival.
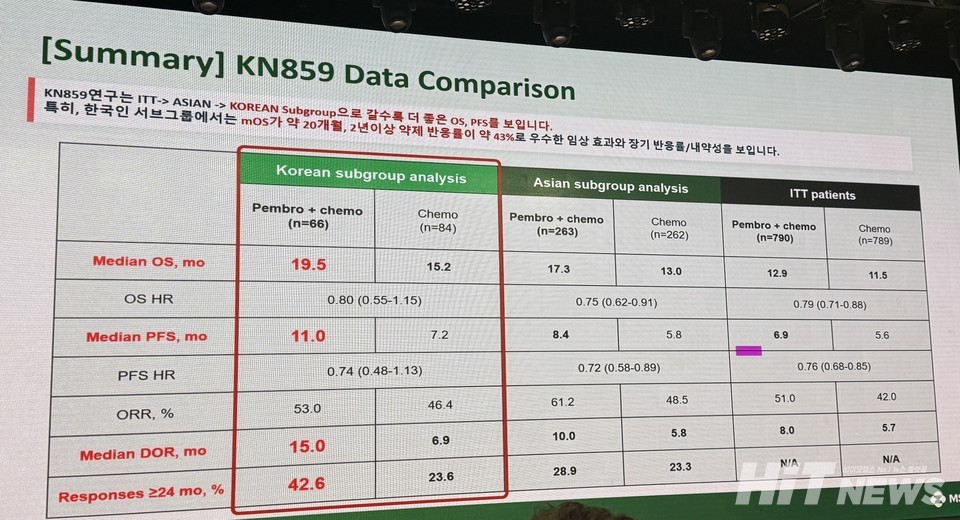
In HER2-negative gastric cancer, the KEYNOTE-859 study demonstrated a two-month improvement in mOS, with a 36% overall survival benefit in patients with a CPS of 10 or higher. Professor Lee added that outcomes were even more favorable among Korean patients.
Reimbursement has also been extended to MSI-H colorectal cancer. MSI-H, a form of genomic instability caused by DNA replication errors, is observed in colorectal, gastric, endometrial, small bowel, and biliary tract cancers.
In the KEYNOTE-177 trial, which compared pembrolizumab with standard chemotherapy in metastatic colorectal cancer, approximately 60% of patients in the control arm crossed over to immunotherapy. Median overall survival reached 77.5 months in the pembrolizumab group, compared with 36.7 months in the control group—more than doubling survival.
“I hope more patients become aware of these effective treatment options and are able to benefit from them,” Professor Lee said.

Professor Min-hwan Kim of Yonsei Cancer Center then presented on “Hope for a New Tomorrow: Keytruda’s Commitment to Underserved Women’s Cancers.”
According to Professor Kim, the most common cancers affecting women in 2023 were breast cancer (21.6%), endometrial cancer (2.9%), and cervical cancer (2.3%). While treatment advances have improved outcomes, five-year survival rates for advanced disease remain low at approximately 30–40%.
Triple-negative breast cancer, which accounts for 15–20% of all breast cancers, often occurs at a younger age and is associated with rapid progression and a high risk of recurrence. Due to the lack of effective targeted therapies, unmet medical needs have remained substantial.
Endometrial cancer, arising from the uterine lining, is associated with risk factors such as obesity, diabetes, and immunodeficiency, with incidence increasing alongside Westernized dietary patterns. Although cervical cancer incidence has declined with HPV vaccination, Professor Kim noted that reduced prevalence has also led to decreased research investment and fewer therapeutic advances.
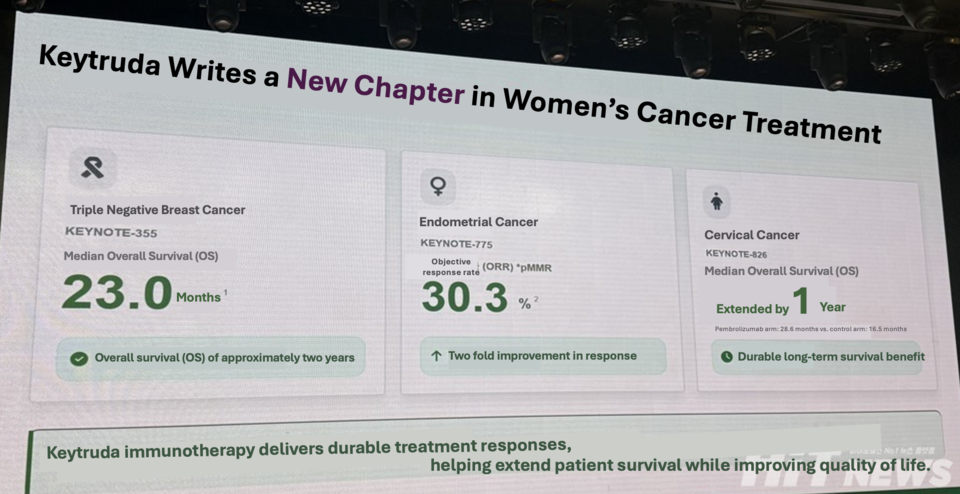
In the KEYNOTE-355 study, Keytruda combination therapy improved median progression-free survival (mPFS) by nearly 1.7-fold compared with chemotherapy alone in PD-L1–high triple-negative breast cancer (9.7 months vs. 5.6 months). In endometrial cancer, the KEYNOTE-775 trial demonstrated a 40% reduction in the risk of disease progression or death in patients with pMMR tumors, while the KEYNOTE-826 study showed a median overall survival extension of more than 12 months in cervical cancer.
Professor Kim emphasized that Keytruda’s key strength lies in its ability to translate response rates into meaningful survival benefits. He added that expanded reimbursement coverage is expected to improve access for patients previously limited by cost or insufficient treatment efficacy.
“While cost-effectiveness is evaluated during reimbursement decisions, reducing the social burden of women’s cancers can also deliver meaningful economic value,” he said. “I hope Keytruda serves as a starting point for improving outcomes in women’s cancers.”
Meanwhile, MSD is conducting clinical trials to demonstrate the non-inferiority of subcutaneous (SC) Keytruda compared with intravenous (IV) administration in indications such as non-small cell lung cancer (NSCLC), as part of its regulatory strategy. In parallel, MSD Oncology plans to expand its antibody–drug conjugate (ADC) pipeline through the clinical development of nine candidate programs.

