Celltrion and LigaChem Biosciences Push Next-Generation Antibody–Drug Conjugates Beyond Padcev’s Benchmark
As the Nectin-4–targeted antibody–drug conjugate (ADC) Padcev (enfortumab vedotin) continues to transform the treatment of urothelial carcinoma—from early to metastatic stages—attention is now turning to Korea’s Celltrion and LigaChem Biosciences, two companies developing next-generation ADCs targeting the same protein.
Nectin-4 is an immunoglobulin-like adhesion molecule expressed at low to moderate levels in normal tissues but highly over-expressed in cancers such as bladder, breast, and pancreatic tumors.
Recent global oncology meetings have underscored the power of Padcev combined with Keytruda (pembrolizumab). In the EV-302 trial presented at ASCO GU 2025, the regimen achieved a median overall survival of 33.8 months, roughly double that of standard platinum-based chemotherapy (15.9 months), with 67.5 % of patients responding and about one-third achieving complete remission. Likewise, the EV-303 peri-operative study presented at ESMO 2025 reported a 24-month event-free survival rate of 74.7 %, cutting the risk of recurrence or death by about 60 %.
Buoyed by these data, Padcev’s worldwide sales have surged from USD 750 million (2022) to USD 1.58 billion (2024), and developer Astellas now projects annual revenue peaks of up to USD 3.4 billion. Against this backdrop, Celltrion and LigaChem Biosciences are positioning their own Nectin-4 ADCs as potential best-in-class challengers.
LigaChem Bio Partners with Nectin Therapeutics on Dual Nectin-4 ADCs
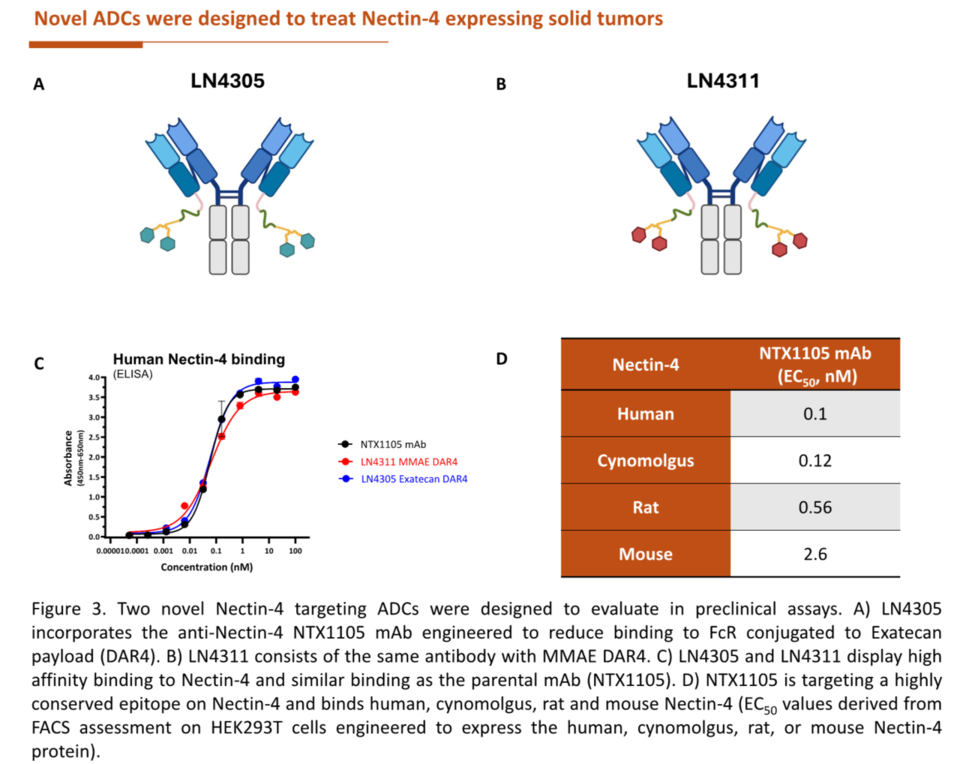
LigaChem Biosciences is co-developing two Nectin-4–targeted ADCs—LN-4305 and LN-4311—with Israeli antibody-discovery firm Nectin Therapeutics. Both use LigaChem’s ConjuALL ADC platform and Nectin Therapeutics’ engineered antibodies that reduce off-target immune interactions, aiming to minimize toxicity.
LN-4305 carries an exatecan payload, while LN-4311 uses monomethyl auristatin E (MMAE), the same cytotoxic agent as Padcev. Presented publicly for the first time at ESMO 2025, both agents showed stronger pre-clinical efficacy and improved safety versus Padcev, including tumor suppression in animal models resistant to Padcev.
Toxicology studies in rats and monkeys indicated >10-fold higher non-toxic doses compared with Padcev, suggesting room for higher clinical exposure. LigaChem believes the candidates could overcome Padcev’s known skin and neurotoxicities while boosting efficacy.
“LN-4305 and LN-4311 delivered deeper, longer tumor control than Padcev in triple-negative breast and non-small cell lung cancer models,” a company spokesperson said. “These data support future expansion to other Nectin-4–high tumors with greater market potential.”
The firm added that IND submission is planned for 2026, followed by global clinical trials and licensing discussions under a shared-revenue structure with Nectin Therapeutics.
Celltrion Advances Phase 1 of K-ADC “CT-P71” with Homegrown Payload
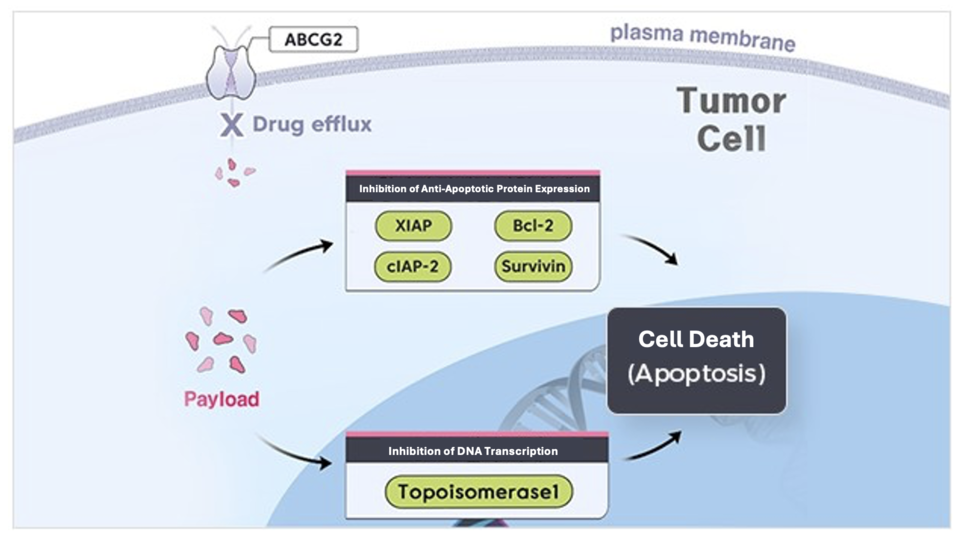
Celltrion is developing CT-P71, a Nectin-4–targeted ADC built around PBX-7016, a novel camptothecin-derived TOP1 inhibitor co-developed with Pinot Bio. The payload induces cancer-cell death by causing DNA damage and inhibiting tumor proliferation, showing potent tumor-growth inhibition and low toxicity in pre-clinical studies.
Unveiled at the 2024 World ADC Conference, CT-P71 received Phase 1 approval from Korea’s MFDS in July 2025. During the Celltrion Science & Innovation Day 2025, the company highlighted CT-P71’s durable activity in organoid and xenograft models, including Padcev-resistant tumors, underscoring its next-generation potential.
The ongoing Phase 1a trial seeks the optimal dose; Phase 1b will identify responsive solid-tumor types, with urothelial carcinoma prioritized for Phase 2. Celltrion also plans to broaden its ADC platform through bispecific and dual-payload constructs.
Analysts note that Celltrion, now ahead of LigaChem in clinical testing, could release interim randomized data within 2025, offering the first clinical glimpse of CT-P71’s competitiveness. The results may define the strategies of both Korean contenders as the race for the Nectin-4 ADC leadership accelerates.

