Dual-Dose NT-I7 to Extend CAR-T Persistence, Boost Outcomes

NeoImmuneTech has begun a Phase 1b clinical trial evaluating repeated administration of its T-cell amplifier NT-I7 after CAR-T therapy in patients with relapsed or refractory large B-cell lymphoma (LBCL). The study aims to address CAR-T’s limitations by sustaining T-cell levels to improve long-term outcomes.
On August 11, NeoImmuneTech (CEO: Patrick Y. Soon-Shiong) announced that the U.S. Food and Drug Administration (FDA) approved the investigator-initiated trial.
CAR-T therapy involves engineering a patient’s own T cells to target cancer cells, but its efficacy is often hindered by the rapid decline of CAR-T cells within weeks of infusion. This drop—typically around Day 14—can lead to lower survival and response rates.
NT-I7 is a long-acting human recombinant interleukin-7 (IL-7) protein designed to enhance T-cell differentiation, proliferation, and recovery in lymphopenic conditions. Engineered for an extended half-life, NT-I7 is in development both as a standalone therapy and in combination with CAR-T, immune checkpoint inhibitors, chemotherapy, and radiotherapy.
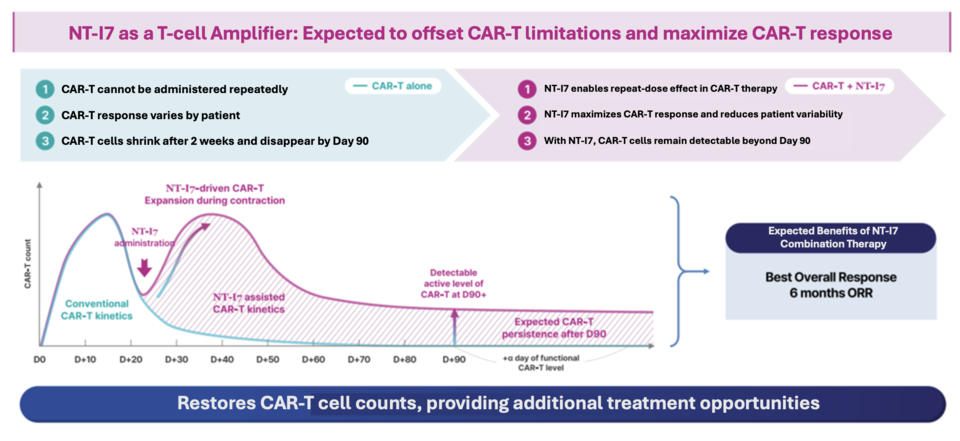
In the earlier NIT-112 trial, high-dose NT-I7 (≥480 μg/kg) reversed CAR-T cell decline, with some patients converting from partial to complete response. The six-month objective response rate (ORR) reached 88%, and doses up to 720 μg/kg were well tolerated. These results prompted a revised dosing strategy.
The new trial will enroll LBCL patients previously treated with Breyanzi or Yescarta. NT-I7 will be administered on Days 10 and 31—earlier and more frequently than the prior single Day 21 dose—to preempt CAR-T decline and reinforce persistence. The 3+3 dose-escalation design will start at 600 μg/kg and increase to 720 μg/kg if safe, before expanding to a 12-patient cohort and later moving to Phase 2 efficacy testing.
Previously, NT-I7 was tested with Keytruda (pembrolizumab) in solid tumors, showing limited ORR gains but a survival benefit. “We consistently see that sufficient T-cell presence correlates with improved survival,” a company spokesperson said.
NeoImmuneTech is also exploring NT-I7 in other T-cell–driven settings, including tumor-infiltrating lymphocyte (TIL) therapy, idiopathic CD4 lymphocytopenia, and acute radiation syndrome (ARS).
In idiopathic CD4 lymphocytopenia, NT-I7 aims to restore immune function by replenishing T cells. For ARS, where radiation rapidly depletes lymphocytes, NT-I7 seeks to normalize counts and improve survival under the FDA’s Animal Rule. Preclinical data in rodents and non-human primates have shown T-cell expansion and survival benefits.
“NT-I7 can be applied to any therapy requiring T-cell amplification,” the spokesperson said. “Our focus starts with CAR-T combinations and will expand into broader T-cell therapies, rare diseases, and radiation damage treatments.”

