Camurus secures exclusive rights for long-acting injectables
Peptron insists its talks with Lilly remain active

Global pharmaceutical leader Eli Lilly has entered a technology transfer agreement with Swedish biotech firm Camurus for its long-acting injectable drug delivery platform. The deal has raised concerns in Korea that Peptron, which had been collaborating with Lilly, may now lose momentum in the competitive drug delivery space.
Peptron had signed a 14-month evaluation agreement with Lilly in October last year to explore the application of its SmartDepot platform to Lilly’s peptide-based drug candidates. Although the specific compound was undisclosed, the collaboration fueled speculation about a potential licensing deal. At its peak, Peptron’s market capitalization exceeded $3.65 billion.
Investor sentiment shifted sharply after Lilly’s announcement on June 3 (U.S. local time), revealing an exclusive global licensing deal with Camurus.
According to the Korea Exchange, Peptron’s shares hit the daily lower limit on June 4, plunging $50.41 to close at $117.63. The company’s market cap fell from $3.92 billion to $2.74 billion, erasing over $1.18 billion in a single day.
Under the new deal, Lilly secured exclusive rights to use Camurus’s FluidCrystal® platform with up to four incretin-based therapies, including key assets such as its dual GIP/GLP-1 agonist, the triple GIP/glucagon/GLP-1 agonist Retatrutide, and the amylin receptor agonist Eloralintide. The agreement also provides Lilly with the option to extend the platform’s use to additional pipeline candidates.
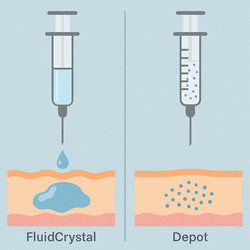
Camurus’s FluidCrystal® is a lipid-based liquid that transforms into a gel-like depot upon injection, enabling sustained drug release for days to months. In contrast, Peptron’s SmartDepot uses PLGA—a U.S. FDA-approved biodegradable polymer—to encapsulate the drug in microspheres, which slowly degrade to maintain therapeutic levels. Though the mechanisms differ, both platforms are designed for long-acting injectable delivery, making Lilly’s choice of Camurus a notable development.
An industry official remarked, “Peptron is one of several companies working with Lilly, not its exclusive partner. The long-acting injectable space remains highly competitive, with ongoing patent battles even among domestic firms. Without concrete evidence of SmartDepot’s superiority, investor confidence may continue to erode.”
In response to market concerns, Peptron issued a statement on June 4 via its website, emphasizing that its evaluation agreement with Lilly remains active. “Technical discussions on long-acting obesity therapeutics are progressing smoothly,” the company stated, adding that “Camurus’s technology operates via an entirely different mechanism and does not constitute direct competition.”
When contacted by Hit News, Peptron responded briefly: “Internal discussions regarding this matter are ongoing. We are unable to provide further comment at this time.”

