Calls for flexible thresholds, pre‑negotiation and provisional coverage

Systemic reforms are needed to ensure that reimbursed drugs retain coverage when used in combination and to facilitate inter‑company negotiations on joint pricing.
On March 17, Member of Parliament Ju‑young Lee hosted a policy forum titled “Improving Cancer Patients’ Access to Combination Therapy,” organized by the Korean Cancer Association and the Korea Research‑based Pharmaceutical Industry Association (KRPIA). Dr. In‑ho Kim, Professor of Medical Oncology at Seoul St. Mary’s Hospital, described combination therapy as a novel approach that overcomes the limitations of existing anticancer treatments.
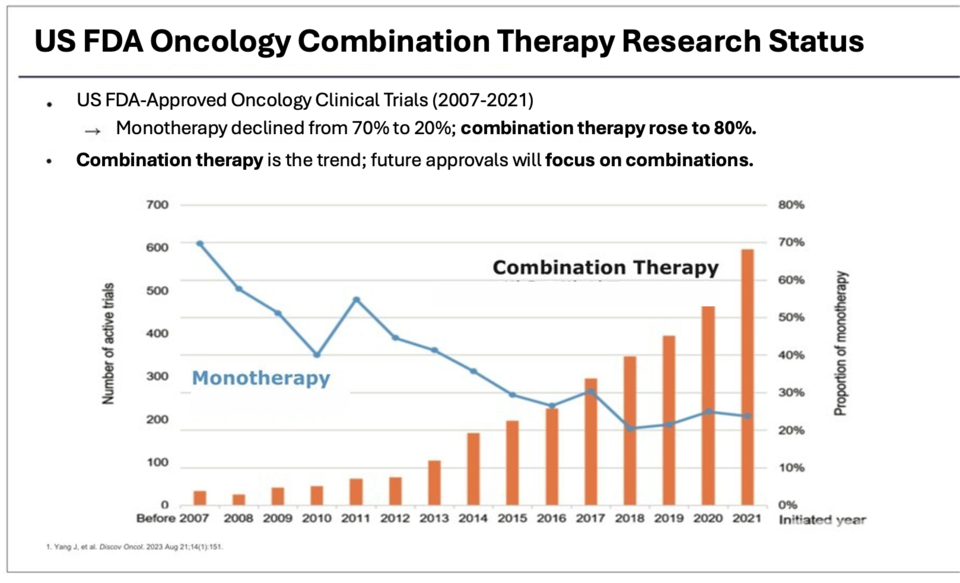
According to Professor Kim, FDA data show that combination‑therapy trials now account for 80% of ongoing oncology research—up from 30% a decade ago—while monotherapy studies have fallen from 70% to 20%. In Korea, more than 70 clinical trials combining approved agents with new drugs have been conducted in the past ten years. These regimens have more than doubled survival in bladder cancer and reduced renal cancer mortality risk by 28%, yet high costs and lengthy approval processes continue to impede patient access.
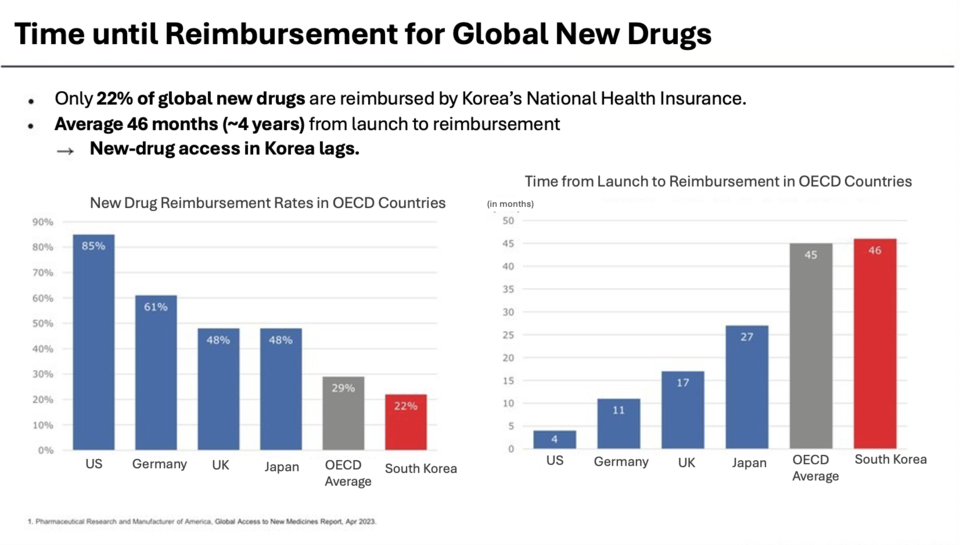
Only 22% of novel oncology drugs approved globally receive reimbursement in Korea, with an average lag of 46 months between market approval and coverage. Professor Kim warned that when a reimbursed drug is paired with a non‑reimbursed agent, it loses its coverage status—driving 87% of cancer patients to consider discontinuing treatment due to cost.
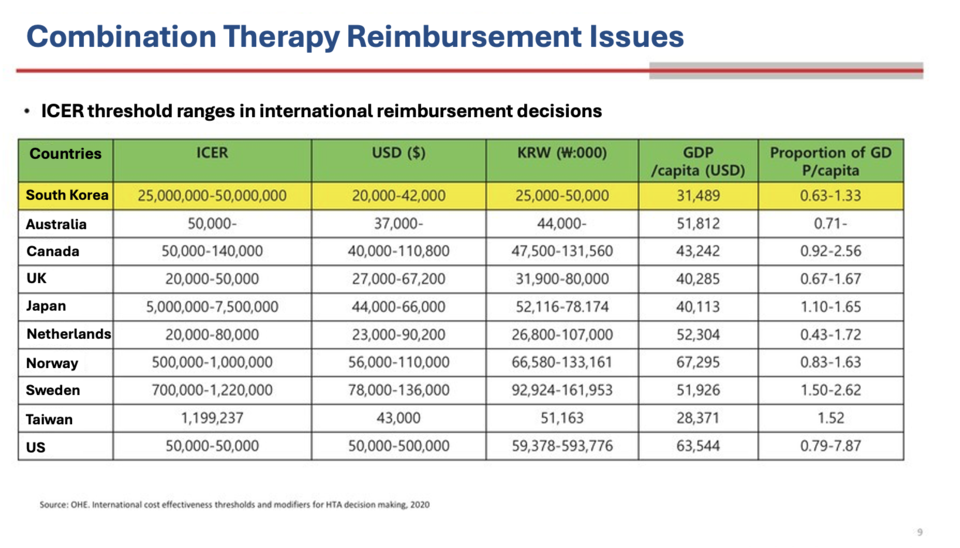
Emeritus Professor Dong‑chul Seo of Chung‑Ang University’s College of Pharmacy noted that combining high‑cost agents typically produces incremental cost‑effectiveness ratios (ICERs) that exceed Korea’s willingness‑to‑pay threshold. Between 2018 and 2022, the median accepted ICER for anticancer drugs in Korea was $27,000 per quality‑adjusted life year (QALY), compared with thresholds up to $92,500 in Sweden. Professor Seo recommended adopting flexible ICER thresholds based on disease severity, unmet need and clinical innovation, and making separate coverage decisions for each agent in a combination regimen.

Professor Seo also highlighted that antitrust regulations prevent price negotiations between manufacturers, delaying joint launches of combination therapies. He proposed that the National Health Insurance Service facilitate pre‑negotiation consultations among companies and mediate pricing discussions. He further advocated for provisional coverage with outcome‑based reimbursement to enable earlier patient access while generating real‑world evidence.
During the panel discussion, Eun‑young Lee, Director of the Korea Patient Coalition, stressed that reimbursement barriers for combination therapies have persisted since the “Dacogen + Venclexta” case a decade ago. “Cancer treatment is inherently long‑term and financially burdensome. Reimbursement criteria must be reformed so patients are not forced to choose between treatment and financial ruin,” she said.
Governmental Response
Hee‑yeon Park, Deputy Director of the Insurance Drug Division at the Ministry of Health and Welfare, acknowledged that stripping coverage from reimbursed drugs in combination regimens is unreasonable. While reforms must guard against unintended consequences, the ministry is consulting with the Health Insurance Review & Assessment Service (HIRA) to develop mediation mechanisms—but offered no firm timeline.
Guk‑hee Kim, Director of Drug Management at HIRA, reported that total pharmaceutical spending rose 15% in 2023, with anticancer drugs accounting for 26% of the increase. She warned that further high‑cost innovations will strain the health insurance budget and underscored the need for strategic treatment sequencing. Kim noted that without cost‑effectiveness data from manufacturers, HIRA lacks the authority to advance reimbursement negotiations.

