Legal Battles Continue Over Usage Patents as Generic Launch Proceeds
South Korean pharmaceutical companies have successfully invalidated an unregistered composition patent for Trajenta Duo, Boehringer Ingelheim’s combination therapy for diabetes. While disputes over usage patents persist, local firms are expected to avoid liability for damages following the launch of generic versions in 2024.
The Korean Intellectual Property Tribunal (KIPT) ruled in favor of South Korean drugmakers—including Kukje Pharma, Shinil Pharmaceutical, Aju Pharm, and Korea Hutex Pharma—in a scope confirmation trial regarding the patent for a “pharmaceutical composition containing BI-1356 and metformin.” The decision effectively curtails Boehringer Ingelheim’s patent rights in South Korea.
The contested patent covered Trajenta Duo (linagliptin/metformin) and was initially set to expire on October 1, 2030. However, it was not included in the Ministry of Food and Drug Safety’s (MFDS) official Green List of pharmaceutical patents. Legal challenges were filed between October and November 2023, coinciding with local firms’ preparations to introduce generics in 2024. This move reflected a broader industry strategy to challenge “hidden patents” that could create legal risks.
South Korean companies accelerated their market entry efforts following the expiration of Trajenta’s compound patent on June 8, 2024. By June 9, 127 generic products from 19 companies were ready for launch. Given that the combined sales of Trajenta monotherapy and Trajenta Duo reached approximately $100 million in 2022, securing early market share was crucial.
However, last-minute concerns arose over unregistered patents not listed with the MFDS. While these do not legally block generic sales, they pose potential risks of litigation, including infringement lawsuits, damage claims, and sales injunctions.
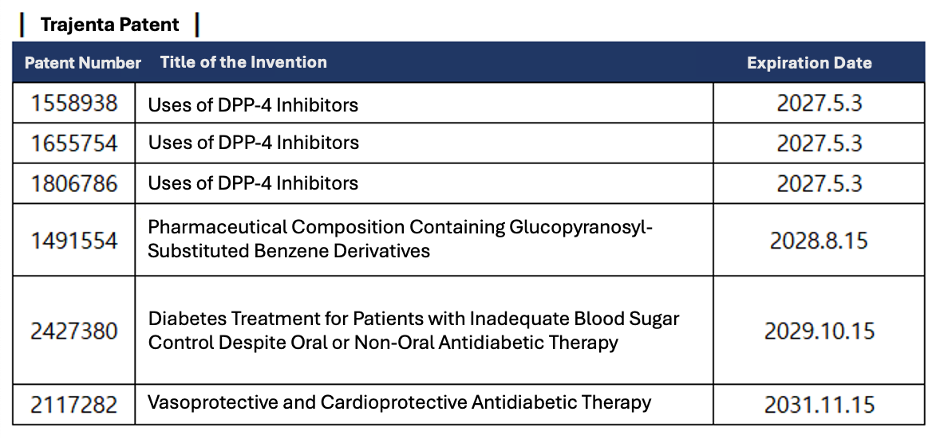
Starting in October 2023, Korean pharmaceutical companies intensified efforts to bypass unregistered patents for Trajenta products, proceeding with their launches in June 2024. Despite preemptive warning notices from Boehringer Ingelheim, local firms pressed forward, calculating that even in the event of litigation, potential damages would be limited and difficult to quantify.
Following the launch, Korean firms engaged in multiple legal battles over Trajenta and Trajenta Duo. At the time of Boehringer Ingelheim’s warnings, at least seven unregistered patents covered Trajenta monotherapy. With the latest ruling favoring local companies, industry estimates suggest around 13 ongoing disputes, with most rulings so far siding with South Korean firms. Notably, a second-instance ruling recently invalidated an unregistered usage patent for Trajenta.
Despite Boehringer Ingelheim’s legal efforts, industry experts consider the risk of South Korean firms losing these cases low. Many of the disputed patents stem from an evergreening strategy, and their complexity is far lower than compound patents. As a result, South Korean pharmaceutical companies are expected to persist in their legal challenges, ensuring continued market access for their generic products.

