Innovative RNA Editing and Therapeutics Gain Global Recognition
Without deep science, solid rationale, and global competitiveness, securing investor confidence is challenging. Sung-wook Lee, CEO of Rznomics, is pioneering RNA-based therapeutics with a strong scientific foundation. In a recent interview with Hit News, he outlined the company’s strategy to expand indications by leveraging its Trans-splicing Ribozyme technology and circular RNA platform, both of which offer distinct advantages and global potential. Building on this foundation, Rznomics secured a pre-IPO investment of $14.2 million last month.
Rznomics' Breakthrough in RNA Editing
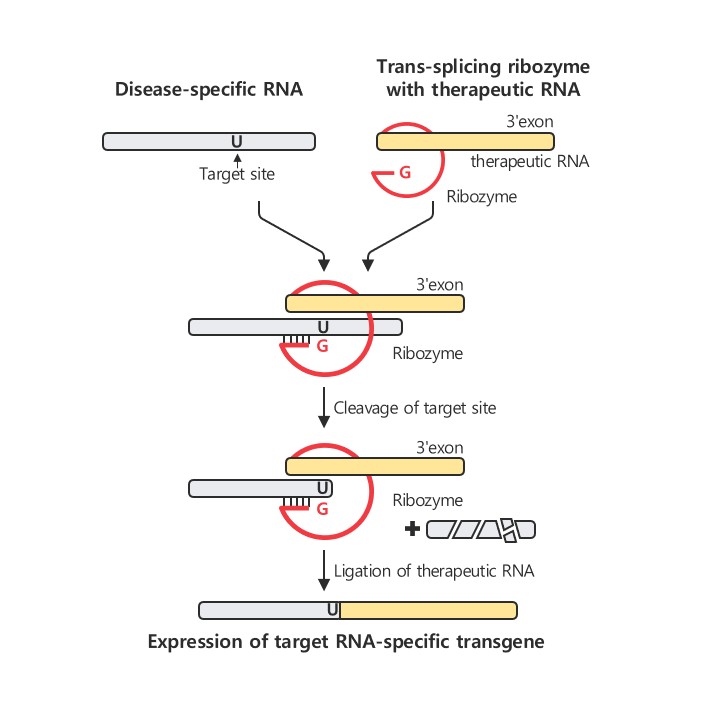
Rznomics is gaining attention with its innovative RNA-based therapeutic platform. Its core technology, Trans-splicing Ribozyme, corrects mutated RNA by replacing it with functional RNA, restoring normal protein production. Unlike DNA-based gene therapies that permanently alter the genome, this approach selectively targets disease-causing RNA, ensuring reversibility and safety.
“AAV vectors have size limitations in genetic material they can carry, but our technology only replaces specific RNA segments requiring correction, mitigating this constraint,” Lee noted. He emphasized that AAV enables stable, long-term RNA expression in non-dividing cells, making it effective for chronic conditions such as CNS and retinal diseases.
Using an analogy, he explained, “Once an AAV vector is introduced into a non-dividing cell, it functions like a factory that continuously produces RNA therapy. A single administration provides lasting therapeutic benefits.”
For transient treatments like cancer, Rznomics employs adenoviruses, which do not integrate into the host genome and act temporarily, making them well-suited for oncology applications.
A major advantage of the Trans-splicing Ribozyme technology is its ability to address multiple genetic mutations with a single drug. “Even if patients have different mutations, our technology can map and correct various RNA errors with one therapeutic agent,” Lee explained.
The technology also prevents overexpression-related side effects. Unlike conventional RNA therapies that risk unintended expression, Rznomics' platform precisely regulates protein production, minimizing adverse effects.
Highlighting its versatility, Lee stated, “Our platform is modular, consisting of components that target specific RNA sequences, facilitate splicing, and deliver therapeutic RNA. This adaptability allows us to develop precise treatments for untreatable rare diseases.”
Expanding to Circular RNA Therapeutics
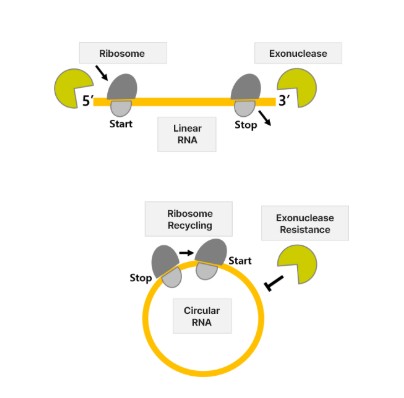
Beyond Trans-splicing Ribozyme, Rznomics is developing a circular RNA (circRNA) platform. Unlike linear RNA, circRNA has a covalently closed-loop structure, making it highly resistant to enzymatic degradation. This ensures prolonged stability and functionality within cells.
“One challenge of RNA therapeutics is immune activation, as the body may recognize exogenous RNA as foreign. While linear RNA requires modifications to suppress immune response, circular RNA naturally evades immune detection, making it safer,” Lee explained.
Rznomics’ approach eliminates unnecessary sequences, ensuring the production of pure circular RNA composed only of the gene of interest. This enhances safety and stability upon cellular introduction.
Circular RNA holds promise in advanced cell and gene therapies, including vaccines. Lee noted its potential role in in vivo CAR-T therapies, where it could serve as a vector for CAR gene delivery, streamlining the treatment process. However, he acknowledged that circular RNA is still in early development and requires further research for commercial viability.
“Several leading research groups are exploring circular RNA for vaccines and in vivo CAR-T applications. Identifying the most competitive use cases remains an ongoing challenge,” he said, adding that large-scale production and process optimization are key hurdles to overcome.
Rznomics Gaining Global Recognition
Rznomics' RNA research has earned recognition from global academia and industry. Its technologies have been featured in Nature Biotechnology and Nature, highlighting their scientific significance. Lee emphasized that such credibility has been instrumental in attracting investment and driving growth.
The company has secured key U.S. FDA designations, including Orphan Drug Designation (ODD) and Fast Track status. “FDA recognition strengthens collaboration opportunities with global pharmaceutical firms and facilitates market entry,” Lee stated.
To accelerate commercialization, Rznomics is actively partnering with major pharmaceutical companies. “These collaborations validate our technology and advance its commercial potential,” Lee noted, underscoring the company’s focus on expanding RNA-based therapeutics across multiple indications.
Beyond innovation, Rznomics is committed to real-world patient applications. “Our goal is to leverage the scalability of our technology to provide better treatment options,” Lee affirmed. He reiterated the company’s ambition to establish itself as a leader in the global RNA therapeutics market with a strong competitive edge.

