Rising Biotech Funding Contrasts with Stagnant Performance in South Korean CROs Amid Overseas Competition
In early 2024, South Korea saw a significant rise in investment in unlisted biotech firms developing new drugs, yet this influx has not spurred similar growth in CRO (Contract Research Organization) revenue and profits. While investor sentiment appears positive, analysts suggest that the industry needs more than funding increases to achieve a recovery, and recommend that CROs look toward international markets to diversify their client base.
Investment in unlisted biotech reached $152 million in the first half of 2024, representing about a quarter of the $614 million allocated to healthcare, according to HitNews. This aligns with historical proportions but is only half of the $289 million invested during the biotech boom of 2021. Notably, it marks a 1.7-fold increase from the $92.5 million raised in the first half of 2023.
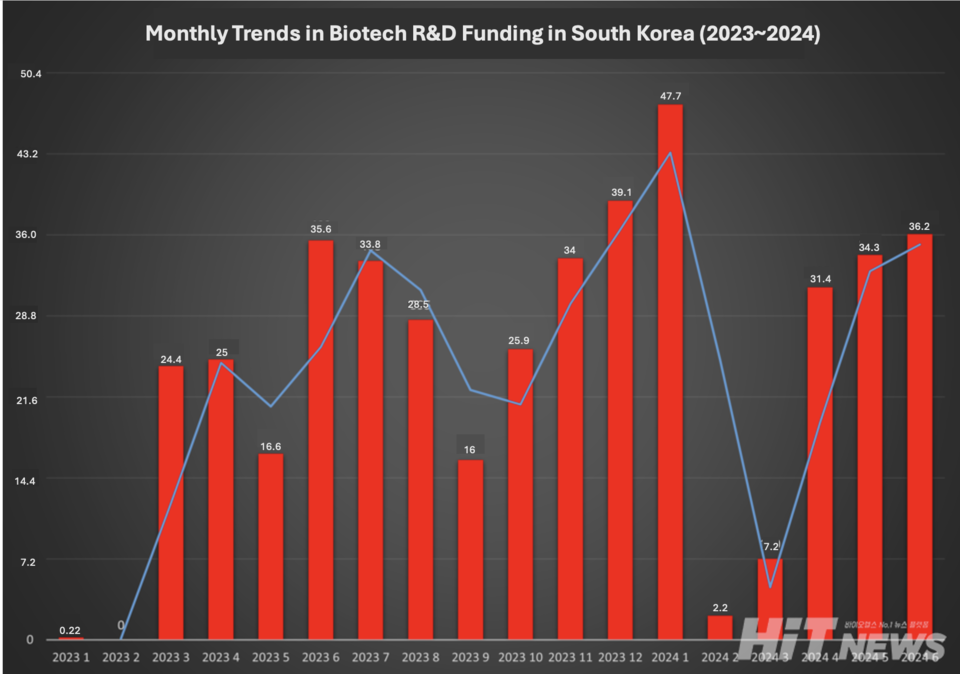
Despite these gains, the CRO sector did not experience the anticipated performance boost. Most funding raised typically flows to CROs for drug development, establishing an expected link between biotech investment and CRO revenue. Yet this year, South Korean CRO performance diverged, with a few clinical CROs showing gains while preclinical CROs lagged behind.
Price Competition Puts Pressure on Smaller Clinical CROs
Clinical CROs like C&R Research, DreamCIS, and Clips BnC, which provide clinical trial services, reported sales growth over last year. C&R Research, however, saw a 20% drop in operating profit, while DreamCIS moved from a loss to a $939,000 profit. Sales increases among CROs did not directly reflect biotech funding recovery, and contract volumes varied, with clinical trial revenue mostly derived from agreements signed two or three years ago.
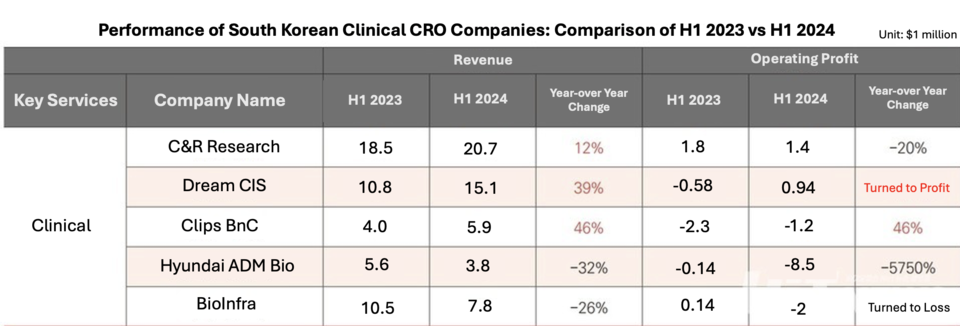
However, this growth in sales is reportedly unrelated to the biotech investment market’s recovery. Total contract volumes for these CROs did not uniformly increase, and CRO sales generally do not immediately reflect new contracts from the current period.
C&R Research’s total contract volume rose from $192.2 million in the first half of last year to $207.2 million this year. According to the company, its revenue structure comprises 70% from new drug clinical trials and 30% from post-marketing surveillance (PMS) or observational studies (OS).
For DreamCIS, total contract volume declined from $31.8 million last year to $26.3 million this year, with around 50% of revenue coming from PMS and OS, and approximately 25% from new drug clinical trials.
In financial disclosures, CRO revenue reflects only the services delivered from the total contract volume, not new contracts signed during the period. A CRO industry representative explained, “The revenue reported in regular disclosures by clinical CROs reflects the biotech investment market from two to three years ago, not the recent year. Revenue is recognized as each milestone in the clinical trial is completed, with payments from the client occurring along these milestones.”
In contrast, clinical CROs Hyundai ADM Bio and BioInfra experienced declines in both sales and operating profits. Industry sources attribute this to intense price competition and policies reducing drug prices, which affect most unlisted clinical CROs as well.
A biotech representative stated, “Since last year, clinical trial quotes from leading South Korean CROs have been noticeably lower, regardless of company size. This prompts companies to entrust trials to larger CROs, where quality may be more assured.”
A clinical CRO business development (BD) representative added, “There are times when I question whether the quotes from other CROs can yield a profit once selling and administrative costs are deducted. With a limited volume of trials available, CROs seem engaged in fierce price competition to capture sales, which favors larger CROs with greater resources.”
Specialist firms, including BioInfra, focusing on bioequivalence (BE) studies, also report struggles due to drug price reduction policies. The stepwise drug pricing system and the “1+3 system,” which limits shared use of clinical trial data, introduced in 2020 and 2021, have led pharmaceutical companies to show less interest in commissioning BE studies for generics.
Thus, the CRO industry’s performance appears minimally impacted by the biotech investment market’s recovery over the past year. Revenue growth in clinical CROs primarily reflects contracts signed years prior, while declines result from intense price competition and the effects of drug price reduction policies.
Preclinical CROs Struggle Amid Biotech Strategy Shifts
In contrast to the mixed results of clinical CROs, most preclinical CROs in South Korea struggled with declining performance. Among the five major KOSDAQ-listed firms, only Woojung Bio showed growth in sales and operating profit in the first half of this year, while others—Corestem ChemOn, HLB bioStep, Biotoxtech, and DT&CRO—saw decreases in both metrics compared to the same period last year.
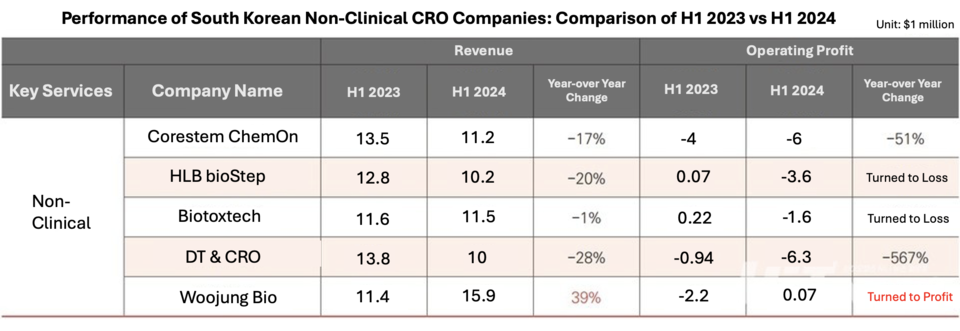
Industry insiders from CROs and biotech companies attribute this unexpected trend, which contradicts previous expectations that "a recovery in biotech investment sentiment would lead to an increase in new drug preclinical development requests," to a shift in biotech companies' strategies driven by lingering market stagnation.
The first major shift has been a reduction in new pipeline development projects. According to industry consensus, “There is a widespread belief that even if a new pipeline is developed, funds for further development may not be available in the future.” A representative from a Series A-stage biotech company said, “We’re not even generating new candidate molecules. Our immediate priority is advancing the lead pipeline to Phase 1, so any preclinical testing we conduct is limited to this pipeline alone.”
The second shift has been a growing preference for using overseas CROs. A preclinical CRO representative explained, “In the past, major overseas preclinical CROs like Charles River, Covance, and WuXi AppTec offered GLP toxicology packages at much higher prices than South Korean firms. Now, they have significantly reduced their package prices to attract Korean biotech clients. Although they are still more expensive, their quality and potential to facilitate global clinical entry make them increasingly appealing.”
This representative added, “Even with a slight recovery in the investment market, biotech sentiment today is entirely different from 2021, when funds were abundant. Back then, domestic preclinical CROs were entrusted with multiple projects to develop various pipelines. However, with the three-year drought in investments, there’s a shift toward focusing resources on the most promising pipeline, often with overseas CRO support to maximize development success.”
Specialization and Overseas Markets Key to CRO Survival
Despite the recovery in the biotech investment market, the preclinical CRO sector continues to struggle due to its heavy reliance on the South Korean market and lack of specialized expertise. This dependency on South Korean demand means performance is highly susceptible to shifts in the Korean biotech landscape. Additionally, a shortage of advanced technical capabilities makes these companies less competitive in terms of both cost and quality against international preclinical CROs. This issue is particularly prevalent among firms offering standardized services like GLP toxicology or non-GLP pharmacokinetics (PK) testing.
Some companies, however, are proactively addressing these challenges. For instance, Humic, an unlisted preclinical CRO, saw its revenue soar from $650,000 last year to $1.0 million by the third quarter of this year. Specializing in humanized mouse models, Humic attributes its success to a team with strong backgrounds in drug development. By combining proprietary models with in-depth knowledge of drug development, they provide consulting on client pipelines, establishing themselves as a CDRO (Contract Development Research Organization).
KeyPrime Research faced difficulties in the first half of this year but is strengthening its specialization as a "primate preclinical testing provider." Currently, approximately 60% of its revenue comes from overseas non-GLP primate PK and DRF (Dose Range Finding) studies. By focusing on primate sourcing and vendor management, KeyPrime has effectively navigated the global primate supply crisis that has persisted since last year. According to a company representative, “Overseas pharmaceutical and biotech clients prioritize quality over price when outsourcing preclinical studies. We start with smaller, simpler studies to build trust and gradually expand contract sizes.”
However, a shortage of skilled personnel remains a challenge. An executive at a listed preclinical CRO noted, “To secure international contracts after developing proprietary technology or specialization, it’s essential to have personnel capable of technical sales. Even for South Korean sales, it’s challenging to find individuals who understand both experimental processes and have a sales mindset.”
The executive added, “Additionally, candidates with foreign language skills often prefer roles in business development (BD) at leading pharmaceutical or biotech firms. The BD shortage has become chronic, making talent acquisition especially difficult for CROs.”

