Broadened coverage for G-CSF injections in neutropenia care
In a recent development anticipated to broaden the landscape of febrile neutropenia treatment, revisions to reimbursement criteria have been unveiled. According to sources within the pharmaceutical industry and statements from the Health Insurance Review & Assessment Service (HIRA) on August 25th, the scope of eligible treatments now encompasses preventive Folfirinox (oxaliplatin + irinotecan + leucovorin + 5-FU) therapy for pancreatic cancer, with G-CSF (granulocyte colony-stimulating factor) injections now eligible for reimbursement.
Of note among the G-CSF injections is the 'Neulasta prefilled syringe,' manufactured by Kyowa Kirin Korea. This specific syringe is utilized in the management of febrile neutropenia in patients undergoing cytotoxic chemotherapy for malignant tumors. The treatment's primary aim is to mitigate the incidence and duration of neutropenia, excluding chronic myeloid leukemia and myelodysplastic syndromes.
Further advancements in reimbursement have been approved for several additional G-CSF injections. 'Neulapeg prefilled syringe' from GC Biopharma, 'Dulastin injection prefilled syringe' from Dong-A-ST, 'Longquex prefilled syringe' from Teva-Handok, and 'Rolontis prefilled syringe' from Hanmi Pharmaceutical have all gained reimbursement approval. These treatments are intended for patients aged 19 and above who have undergone cytotoxic chemotherapy for solid tumors and malignant lymphomas, seeking to reduce the duration of severe neutropenia. Eligibility for coverage extends to specific cancer types and chemotherapy regimens outlined in the reimbursement criteria.
Neutropenia, a condition marked by an abnormal reduction in neutrophil proportions—usually comprising 50-70% of white blood cells—often arises in cancer patients undergoing repeated chemotherapy cycles, leading to a common decrease in neutrophil counts.
Acknowledging this context, the Hematology Society has actively advocated for the expansion of reimbursement criteria, specifically in relation to prophylactic G-CSF injections administered within the 'Folfirinox' regimen for pancreatic cancer. It is projected that this expansion will take effect in the upcoming month.
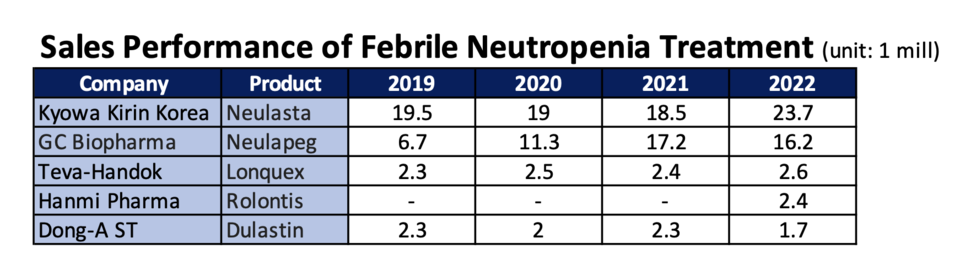
Reflecting on market dynamics over the past year, changes have manifested through distributor transitions and the introduction of novel drugs. IQVIA data provides insights into the performance of key neutropenia treatment drugs up until the previous year. Notably, 'Neulasta' achieved sales of $23.7 million (31.4 billion won). A partnership between Kyowa Kirin and Boryung Pharmaceuticals facilitated joint distribution of 'Neulapeg,' resulting in sales growth from $18.5 million (24.5 billion won) in 2021 to $23.7 million (31.4 billion won) in 2022, exemplifying the partnership's efficacy.
Within this landscape, Boryung Pharmaceuticals disengaged from GC Biopharma and aligned with Kyowa Kirin, while Jeil Pharmaceuticals filled the gap left by Boryung, distributing 'Neulapeg.' Consequently, 'Neulapeg' experienced a minor sales dip from $17.2 million (22.8 billion won) in 2021 to $16.2 million (21.5 billion won) in 2022.
Among the newcomers, the recently launched 'Rolontis' garnered sales of $2.4 million (3.2 billion won) in 2022, outpacing Dong-A ST's 'Dulastin' and securing fourth place. 'Longquex' witnessed sales growth of $2.6 million (3.5 billion won), resulting in a marginal variance, inciting intrigue regarding potential shifts in rankings for the current year.
Meanwhile, the expansion of reimbursement for G-CSF injections brings about adjustments in upper limits. Neulasta's ceiling will decrease from $588 (778,334 won) to $577.50 (764,324 won), while Neulapeg's upper limit will be recalibrated from $428.48 (567,089 won) to $427.62 (565,952 won). These changes are slated for implementation beginning September 1st.

