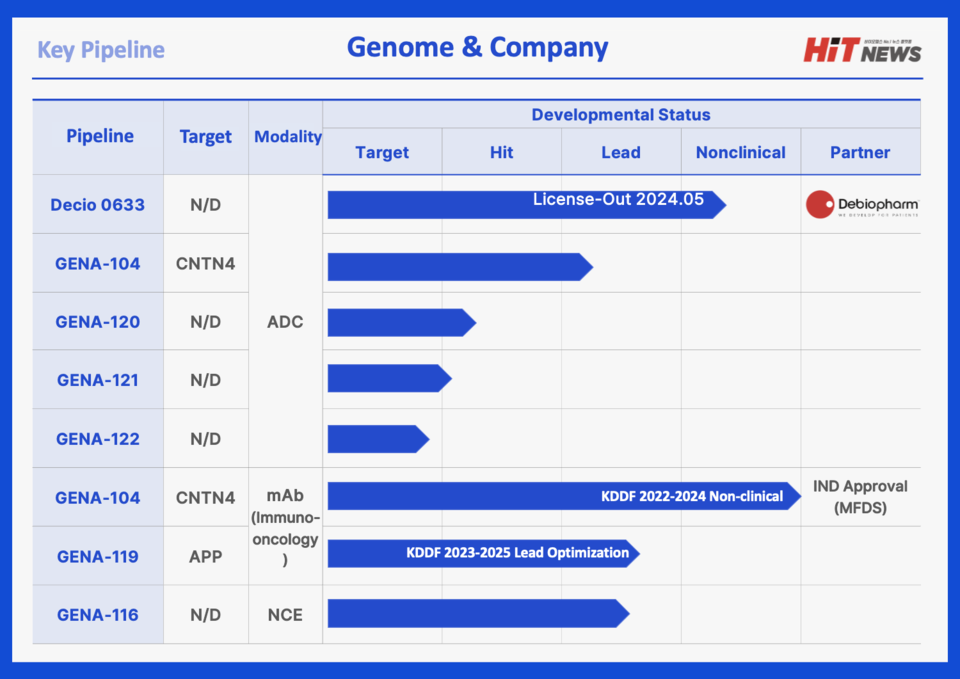
Founded in 2015, Genome & Company is a biotechnology firm specializing in immuno-oncology with a focus on developing first-in-class antibody-drug conjugates (ADCs). The company leverages its proprietary GNOCLE platform to discover novel cancer targets and develop them as either standalone antibodies or ADCs, forming a pipeline aimed at global out-licensing.
The company’s lead candidate, GENA-104, is an immune checkpoint inhibitor that targets CNTN4, a molecule involved in tumor immune evasion. GENA-104 restores T-cell activity by blocking the CNTN4-APP pathway. Notably, CNTN4 is highly expressed in various solid tumors and is mutually exclusive with PD-L1 expression, indicating potential in patients unresponsive to PD-1/PD-L1 therapies.
In February 2024, Genome & Company licensed GENA-104 to UK-based Ellipses Pharma, with plans to initiate Phase 1 clinical trials in both the UK and the U.S.
Building on this success, the company developed GENA-104 ADC, which conjugates the GENA-104 antibody to a cytotoxic payload. The ADC selectively targets CNTN4-expressing tumor cells, delivers the drug intracellularly, and induces cell death. Preclinical studies demonstrated a 100% tumor suppression rate, supported by strong internalization and antitumor activity in animal models.
In May 2024, GENA-104 ADC was out-licensed to Swiss biotech Debiopharm in a deal worth up to $430 million, including a $5.1 million upfront payment.
Beyond the GENA-104 program, Genome & Company is advancing three additional immuno-oncology candidates—GENA-120, GENA-121, and GENA-122—currently in preclinical development via the GNOCLE platform. These assets are being explored as both ADCs and immune antibodies, reinforcing the company’s strategy to expand its first-in-class oncology pipeline.